Empirical Formulae in Chemistry
In chemistry, the empirical formula of a chemical compound is the simplest positive integer ratio of atoms present in a compound. A simple example of this concept is that the empirical formula of hydrogen peroxide, or H2O2, would simply be HO. Glucose (C6H12O6), ribose (C5H10O5), acetic acid (C2H4O2), and formaldehyde (CH2O) all have different molecular formulas but the same empirical formula: CH2O. This is the actual molecular formula for formaldehyde, but acetic acid has double the number of atoms, ribose has five times the number of atoms, and glucose has six times the number of atoms.
In this article we are not discussing the above " Empirical formulas ". There is another kind of " Empirical formulas " where the constants are determined by experiments, rather than derived. The dimesions on the left side or in the right sides may or may not match, case to case basis.
1) Slater's rule
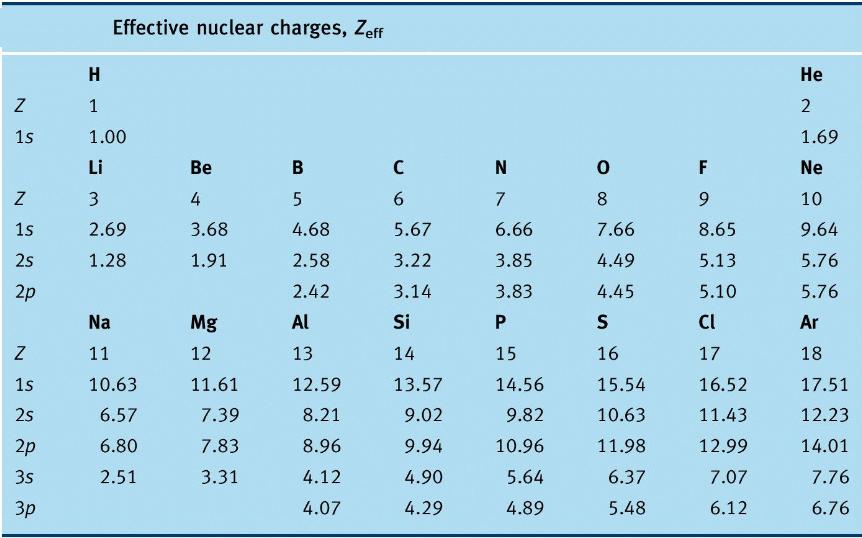
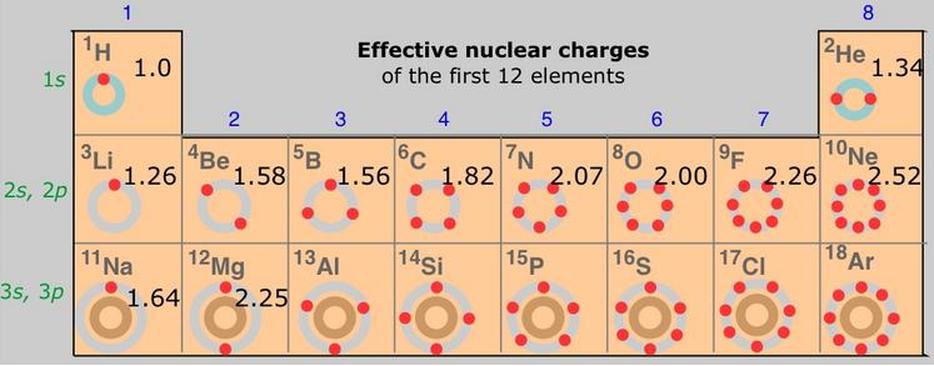
In quantum chemistry, Slater's rules provide numerical values for the effective nuclear charge concept. In a many-electron atom, each electron is said to experience less than the actual nuclear charge owing to shielding or screening by the other electrons. For each electron in an atom, Slater's rules provide a value for the screening constant, denoted by s, S, or s, which relates the effective and actual nuclear charges as
Zeffective = Z - s ( sigma )
The rules were devised semi-empirically by John C. Slater and published in 1930.
Revised values of screening constants based on computations of atomic structure by the Hartree–Fock method were obtained by Enrico Clementi et al in the 1960s
Steps to follow -
1.1) Write the electron configuration for the atom using the following design;
(1s)(2s,2p)(3s,3p) (3d) (4s,4p) (4d) (4f) (5s,5p)
1.2) Any electrons to the right of the electron of interest contributes no shielding. (Approximately correct statement.)
1.3) All other electrons in the same group as the electron of interest shield to an extent of 0.35 nuclear charge units
1.4) If the electron of interest is an s or p electron: All electrons with one less value of the principal quantum number shield to an extent of 0.85 units of nuclear charge. All electrons with two less values of the principal quantum number shield to an extent of 1.00 units.
1.5) If the electron of interest is an d or f electron: All electrons to the left shield to an extent of 1.00 units of nuclear charge.
1.6) Sum the shielding amounts from steps 2 through 5 and subtract from the nuclear charge value to obtain the effective nuclear charge.
Examples:
Calculate Z* for a valence electron in fluorine.
(1s2)(2s2,2p5)
Rule 2 does not apply; 0.35 · 6 + 0.85 · 2 = 3.8
Z* = 9 – 3.8 = 5.2 for a valence electron.
-
Calculate Z* for a 6s electron in Platinum.
(1s2)(2s2,2p6)(3s2,3p6) (3d10) (4s2,4p6) (4d10) (4f14) (5s2,5p6) (5d8) (6s2)
Rule 2 does not apply; 0.35 · 1 + 0.85 · 16 + 60 · 1.00 = 73.95
Z* = 78 – 73.95 = 4.15 for a valence electron.
Shielding
The first ionization energy for hydrogen is 1310 kJ·mol–1 while the first ionization energy for lithium is 520 kJ·mol–1. The IE for lithium is lower for two reasons-
1.7 ) The average distance from the nucleus for a 2s electron is greater than a 1s electron;
1.8 ) The 2s1 electron in lithium is repelled by the inner core electrons, so the valence electron is easily removed.
The inner core electrons shield the valence electron from the nucleus so the outer most electron only experiences an effective nuclear charge. In the case of the lithium the bulk of the 1s electron density lies between the nucleus and the 2s1 electron. So the valence electron `sees' the sum of the charges or approximately +1. In reality the charge the valence electron experiences is greater than 1 because the radial distribution show there is some probability of finding the 2s electron close to the nucleus.
In this article we are not discussing the above " Empirical formulas ". There is another kind of " Empirical formulas " where the constants are determined by experiments, rather than derived. The dimesions on the left side or in the right sides may or may not match, case to case basis.
1) Slater's rule
In quantum chemistry, Slater's rules provide numerical values for the effective nuclear charge concept. In a many-electron atom, each electron is said to experience less than the actual nuclear charge owing to shielding or screening by the other electrons. For each electron in an atom, Slater's rules provide a value for the screening constant, denoted by s, S, or s, which relates the effective and actual nuclear charges as
Zeffective = Z - s ( sigma )
The rules were devised semi-empirically by John C. Slater and published in 1930.
Revised values of screening constants based on computations of atomic structure by the Hartree–Fock method were obtained by Enrico Clementi et al in the 1960s
Steps to follow -
1.1) Write the electron configuration for the atom using the following design;
(1s)(2s,2p)(3s,3p) (3d) (4s,4p) (4d) (4f) (5s,5p)
1.2) Any electrons to the right of the electron of interest contributes no shielding. (Approximately correct statement.)
1.3) All other electrons in the same group as the electron of interest shield to an extent of 0.35 nuclear charge units
1.4) If the electron of interest is an s or p electron: All electrons with one less value of the principal quantum number shield to an extent of 0.85 units of nuclear charge. All electrons with two less values of the principal quantum number shield to an extent of 1.00 units.
1.5) If the electron of interest is an d or f electron: All electrons to the left shield to an extent of 1.00 units of nuclear charge.
1.6) Sum the shielding amounts from steps 2 through 5 and subtract from the nuclear charge value to obtain the effective nuclear charge.
Examples:
Calculate Z* for a valence electron in fluorine.
(1s2)(2s2,2p5)
Rule 2 does not apply; 0.35 · 6 + 0.85 · 2 = 3.8
Z* = 9 – 3.8 = 5.2 for a valence electron.
-
Calculate Z* for a 6s electron in Platinum.
(1s2)(2s2,2p6)(3s2,3p6) (3d10) (4s2,4p6) (4d10) (4f14) (5s2,5p6) (5d8) (6s2)
Rule 2 does not apply; 0.35 · 1 + 0.85 · 16 + 60 · 1.00 = 73.95
Z* = 78 – 73.95 = 4.15 for a valence electron.
Shielding
The first ionization energy for hydrogen is 1310 kJ·mol–1 while the first ionization energy for lithium is 520 kJ·mol–1. The IE for lithium is lower for two reasons-
1.7 ) The average distance from the nucleus for a 2s electron is greater than a 1s electron;
1.8 ) The 2s1 electron in lithium is repelled by the inner core electrons, so the valence electron is easily removed.
The inner core electrons shield the valence electron from the nucleus so the outer most electron only experiences an effective nuclear charge. In the case of the lithium the bulk of the 1s electron density lies between the nucleus and the 2s1 electron. So the valence electron `sees' the sum of the charges or approximately +1. In reality the charge the valence electron experiences is greater than 1 because the radial distribution show there is some probability of finding the 2s electron close to the nucleus.
2 ) Moseley's Law
This law relates to the frequency of the spectral lines of the characteristic X-radiation of a chemical element to its atomic number. This law was experimentally established by H. Moseley in 1913. According to Moseley’s law, the square root of the frequency v of a spectral line of the characteristic radiation of an element is a linear function of its atomic number Z:
3 ) Trouton's law
Trouton’s rule states that the entropy of vaporization is almost the same value, about 85–88 J K-1 mol-1, for various kinds of liquids at their boiling points.The entropy of vaporization is defined as the ratio between the enthalpy of vaporization and the boiling temperature. It is named after Frederick Thomas Trouton.
It also can be expressed as Delta S = 10.5 R
Or better approximation will be S = 4.5 R + R Ln T where T is Temperature in Kelvin. if T = 400 K, we find the original formulation for Trouton's rule.
Trouton’s rule is valid for many liquids; for instance, the entropy of vaporization of toluene is 87.30 J K-1 mol-1, that of benzene is 89.45 J K-1 mol-1, and that of chloroform is 87.92 J K-1 mol-1. Because of its convenience, the rule is used to estimate the enthalpy of vaporization of liquids whose boiling points are known.
The rule, however, has some exceptions. For example, the entropies of vaporization of water, ethanol, and formic acid are far from the predicted values. The entropy of vaporization of XeF6 at its boiling point has the extraordinarily high value of 136.9 J K-1 mol-1. The characteristic of those liquids to which Trouton’s rule cannot be applied is their special interaction between molecules such as hydrogen bonding.
The entropy of vaporization of water and ethanol shows positive deviance from the rule; this is because the hydrogen bonding in the liquid phase lessens the entropy of the phase. In contrast, the entropy of vaporization of formic acid has negative deviance. This fact indicates the existence of an orderly structure in the gas phase; it is known that formic acid forms a dimer structure even in the gas phase. Negative deviance can also occur as a result of a small gas phase entropy owing to a low population of excited rotational states in the gas phase, particularly in small molecules such as methane - a small moment of inertia I giving rise to a large rotational constant B, with correspondingly widely separated rotational energy levels and, via Maxwell–Boltzmann distribution, a small population of excited rotational states and hence a low rotational entropy.
4 ) Einstein-Debey equation (Dulong & Petit)
The specific heat of copper is 0.093 cal/gm K (.389 J/gm K) and that of lead is only 0.031 cal/gm K(.13 J/gm K). Why are they so different? The difference is mainly because it is expressed as energy per unit mass; if you express it as energy per mole, they are very similar.
In 1819 by French physicists Pierre Louis Dulong and Alexis Thérèse Petit, states the classical expression for the molar specific [heat capacity] of certain chemical elements. Experimentally the two scientists had found that the heat capacity per weight (the mass-specific heat capacity) for a number of elements was close to a constant value, after it had been multiplied by a number representing the presumed relative atomic weight of the element. These atomic weights had shortly before been suggested by John Dalton and modified by Jacob Berzelius.
In modern terms, Dulong and Petit found that the heat capacity of a mole of many solid elements is about 3R, where R is the modern constant called the universal gas constant. Dulong and Petit were unaware of the relationship with R, since this constant had not yet been defined from the later kinetic theory of gases. The value of 3R is about 25 joules per kelvin, and Dulong and Petit essentially found that this was the heat capacity of certain solid elements per mole of atoms they contained.
Dulong and Petit gave an Empirical Law for molar specific heat of Solids. The Dulong–Petit law, a thermodynamic rule proposed in 1819 by French physicists Pierre Louis Dulong and Alexis Thérèse Petit, states the classical expression for the molar specific heat capacity of a crystal. Experimentally the two scientists had found that the heat capacity per weight (the mass-specific heat capacity) for a number of substances became close to a constant value, after it had been multiplied by number-ratio representing the presumed relative atomic weight of the substance. These atomic weights had shortly before been suggested by Dalton.
In modern terms, Dulong and Petit found that the heat capacity of a mole of many solid substances is about 3R, where R is the modern constant called the universal gas constant. Dulong and Petit were unaware of the relationship with R, since this constant had not yet been defined from the later kinetic theory of gases. The value of 3R is about 25 joules per kelvin ( Close to 6 Calories per Kelvin ), and Dulong and Petit essentially found that this was the heat capacity of crystals, per mole of atoms they contained.
The modern theory of the heat capacity of solids states that it is due to lattice vibrations in the solid, and was first derived in crude form from this assumption by Albert Einstein, in 1907. The Einstein solid model thus gave for the first time a reason why the Dulong–Petit law should be stated in terms of the classical heat capacities for gases.
Einstein's oscillator treatment of specific heat gave qualitative agreement with experiment and gave the correct high temperature limit (the Law of Dulong and Petit). The quantitative fit to experiment was improved by Debye's recognition that there was a maximum number of modes of vibration in a solid. He pictured the vibrations as standing wave modes in the crystal, similar to the electromagnetic modes in a cavity which successfully explained blackbody radiation. The density of states for these modes, which are called "phonons", is of the same form as the photon density of states in a cavity.
In thermodynamics and solid state physics, the Debye model is a method developed by Peter Debye in 1912 for estimating the phonon contribution to the specific heat (heat capacity) in a solid. It treats the vibrations of the atomic lattice (heat) as phonons in a box, in contrast to the Einstein model, which treats the solid as many individual, non-interacting quantum harmonic oscillators. The Debye model correctly predicts the low temperature dependence of the heat capacity, which is proportional to T3 ( T Cube )
5 ) Reynolds number
In fluid mechanics, the Reynolds number (Re) is a dimensionless quantity that is used to help predict similar flow patterns in different fluid flow situations. The concept was introduced by George Gabriel Stokes in 1851, but the Reynolds number is named after Osborne Reynolds (1842–1912), who popularized its use in 1883
The Reynolds number is defined as the ratio of inertial forces to viscous forces and consequently quantifies the relative importance of these two types of forces for given flow conditions
Reynolds numbers frequently arise when performing scaling of fluid dynamics problems, and as such can be used to determine dynamic similitude between two different cases of fluid flow. They are also used to characterize different flow regimes within a similar fluid, such as laminar or turbulent flow:
laminar flow occurs at low Reynolds numbers, where viscous forces are dominant, and is characterized by smooth, constant fluid motion;
turbulent flow occurs at high Reynolds numbers and is dominated by inertial forces, which tend to produce chaotic eddies, vortices and other flow instabilities.
In practice, matching the Reynolds number is not on its own sufficient to guarantee similitude. Fluid flow is generally chaotic, and very small changes to shape and surface roughness can result in very different flows. Nevertheless, Reynolds numbers are a very important guide and are widely used.
R = Inertial Forces / Viscous Forces = Rho v L by Mu
Inertial Force = Rho (V square) ( L square )
Viscous Force = Mu V L
where
Rho = is the density of the fluid (kg/m³).
v = is the kinematic viscosity mu/rho (m²/s)
L = is a characteristic linear dimension, (travelled length of the fluid; hydraulic diameter when dealing with river systems) (m)
Mu = is the dynamic viscosity of the fluid (Pa·s or N·s/m² or kg/(m·s))
6 ) Raoult's law
Raoult's law states that the vapor pressure of a solvent above a solution is equal to the vapor pressure of the pure solvent at the same temperature scaled by the mole fraction of the solvent present
In the 1880s ( 1882 ), French chemist François-Marie Raoult discovered that when a substance is dissolved in a solution, the vapor pressure of the solution will generally decrease. This observation depends on two variables:
The mole fraction of the amount of dissolved solute present and the original vapor pressure (pure solvent).
At any given temperature for a particular solid or liquid, there is a pressure at which the vapor formed above the substance is in dynamic equilibrium with its liquid or solid form. This is the vapor pressure of the substance at that temperature. At equilibrium, the rate at which the solid or liquid evaporates is equal to the rate that the gas is condensing back to its original form. All solids and liquids have a vapor pressure, and this pressure is constant regardless of how much of the substance is present.
If you look review the concepts of colligative properties, you will find that adding a solute lowers vapor pressure because the additional solute particles will fill the gaps between the solvent particles and take up space. This means less of the solvent will be on the surface and less will be able to break free to enter the gas phase, resulting in a lower vapor pressure.
In mixtures showing a positive deviation from Raoult's Law, the vapour pressure of the mixture is always higher than you would expect from an ideal mixture.
Also you can have mixtures with vapour pressures which are less than would be expected by Raoult's Law. In some cases, the deviations are small, but in others they are much greater giving a minimum value for vapour pressure lower than that of either pure component.
Let Take a binary solution with have components A and B. If the force of attraction between molecular of and A and B in the solution are stronger than that of between A — A and B — B, then the tendency of escaping of molecules A—B from the solution becomes less than that of pure liquids. The total pressure of the solution will be lower than the corresponding vapour pressure of ideal solution of the same component A and B. This type of solution shows negative deviation from Raoult's law. The negative deviation of solution have been shown in figure. Some energy released when we mix both solutions. This reaction is exothermic reaction. For Exothermic reaction delta H is always negative .
7 ) Variation of viscosity with temperature
With an increase in temperature, there is typically an increase in the molecular interchange as molecules move faster in higher temperatures.
The gas viscosity will increase with temperature. According to the kinetic theory of gases, viscosity should be proportional to the square root of the absolute temperature, in practice, it increases more rapidly.
In a liquid there will be molecular interchange similar to those developed in a gas, but there are additional substantial attractive, cohesive forces between the molecules of a liquid (which are much closer together than those of a gas). Both cohesion and molecular interchange contribute to liquid viscosity.
The impact of increasing the temperature of a liquid is to reduce the cohesive forces while simultaneously increasing the rate of molecular interchange.
The former effect causes a decrease in the shear stress while the latter causes it to increase. The result is that liquids show a reduction in viscosity with increasing temperature. With high temperatures, viscosity increases in gases and decreases in liquids, the drag force will do the same.
The impact of increasing temperature will be to slow down the sphere in gases and to accelerate it in liquids. When you consider a liquid at room temperature, the molecules are tightly bound together by attractive inter-molecular forces (e.g. Van der Waal forces).
It is these attractive forces that are responsible for the viscosity since it is difficult for individual molecules to move because they are tightly bound to their neighbors.
The increase in temperature causes the kinetic or thermal energy to increase and the molecules become more mobile.
The attractive binding energy is reduced and therefore the viscosity is reduced. If you continue to heat the liquid the kinetic energy will exceed the binding energy and molecules will escape from the liquid and it can become a vapor.
So the temperature dependence of liquid viscosity is the phenomenon by which liquid viscosity tends to decrease (or, alternatively, its fluidity tends to increase) as its temperature increases. This can be observed, for example, by watching how cooking oil appears to move more fluidly upon a frying pan after being heated by a stove.
8 ) Variation of surface tension with temperature
Surface tension is dependent on temperature. For that reason, when a value is given for the surface tension of an interface, temperature must be explicitly stated. The general trend is that surface tension decreases with the increase of temperature, reaching a value of 0 at the critical temperature. For further details see the Eötvös rule below. There are only empirical equations to relate surface tension and temperature:
9 ) Debye-Huckel-Onsager theory
10 ) Liquid drop model of Nucleus
In nuclear physics, description of atomic nuclei formulated (1936) by Niels Bohr and used (1939) by him and John A. Wheeler to explain nuclear fission. According to the model, the nucleons (neutrons and protons) behave like the molecules in a drop of liquid. If given sufficient extra energy (as by the absorption of a neutron), the spherical nucleus may be distorted into a dumbbell shape and then split at the neck into two nearly equal fragments, releasing energy. Although inadequate to explain all nuclear phenomena, the theory underlying the model provides excellent estimates of average properties of nuclei.
The semi-empirical mass formula (SEMF) (sometimes also called Weizsäcker's formula, or the Bethe-Weizsäcker formula, or the Bethe-Weizsäcker mass formula to distinguish it from the Bethe–Weizsäcker process) is used to approximate the mass and various other properties of an atomic nucleus from its number of protons and neutrons. As the name suggests, it is based partly on theory and partly on empirical measurements. The theory is based on the liquid drop model proposed by George Gamow, which can account for most of the terms in the formula and gives rough estimates for the values of the coefficients. It was first formulated in 1935 by German physicist Carl Friedrich von Weizsäcker, and although refinements have been made to the coefficients over the years, the structure of the formula remains the same today.
The SEMF gives a good approximation for atomic masses and several other effects, but does not explain the appearance of magic numbers of protons and neutrons, and the extra binding-energy and measure of stability that are associated with these numbers of nucleons.
10 ) Nuclear Shell Model
Maria Goeppert Mayer, who made important discoveries about nuclear structure, is one of only two women to have won the Nobel Prize in physics.
In August 1948, Goeppert Mayer published her first paper detailing the evidence for the nuclear shell model, which accounts for many properties of atomic nuclei.
During her time at Chicago and Argonne in the late 1940s, Goeppert Mayer developed a mathematical model for the structure of nuclear shells, which she published in 1950. Her model explained why certain numbers of nucleons in an atomic nucleus result in particularly stable configurations. These numbers are what Eugene Wigner called magic numbers: 2, 8, 20, 28, 50, 82, and 126. Enrico Fermi provided a critical insight by asking her: "Is there any indication of spin orbit coupling?" She realised that this was indeed the case, and postulated that the nucleus is a series of closed shells and pairs of neutrons and protons tend to couple together. She described the idea as follows:
Think of a room full of waltzers. Suppose they go round the room in circles, each circle enclosed within another. Then imagine that in each circle, you can fit twice as many dancers by having one pair go clockwise and another pair go counterclockwise. Then add one more variation; all the dancers are spinning twirling round and round like tops as they circle the room, each pair both twirling and circling. But only some of those that go counterclockwise are twirling counterclockwise. The others are twirling clockwise while circling counterclockwise. The same is true of those that are dancing around clockwise: some twirl clockwise, others twirl counterclockwise.
Three German scientists, Otto Haxel, J. Hans D. Jensen, and Hans Suess, were also working on solving the same problem, and arrived at the same conclusion independently. Their results were announced in the issue of the Physical Review before Goeppert Mayer 's announcement in June 1949. Afterwards, she collaborated with them. Hans Jensen co-authored a book with Goeppert Mayer in 1950 titled Elementary Theory of Nuclear Shell Structure. In 1963, Goeppert Mayer, Jensen, and Wigner shared the Nobel Prize for Physics "for their discoveries concerning nuclear shell structure." She was the second and most recent female Nobel laureate in physics, after Marie Curie.
In nuclear physics, description of atomic nuclei formulated (1936) by Niels Bohr and used (1939) by him and John A. Wheeler to explain nuclear fission. According to the model, the nucleons (neutrons and protons) behave like the molecules in a drop of liquid. If given sufficient extra energy (as by the absorption of a neutron), the spherical nucleus may be distorted into a dumbbell shape and then split at the neck into two nearly equal fragments, releasing energy. Although inadequate to explain all nuclear phenomena, the theory underlying the model provides excellent estimates of average properties of nuclei.
The semi-empirical mass formula (SEMF) (sometimes also called Weizsäcker's formula, or the Bethe-Weizsäcker formula, or the Bethe-Weizsäcker mass formula to distinguish it from the Bethe–Weizsäcker process) is used to approximate the mass and various other properties of an atomic nucleus from its number of protons and neutrons. As the name suggests, it is based partly on theory and partly on empirical measurements. The theory is based on the liquid drop model proposed by George Gamow, which can account for most of the terms in the formula and gives rough estimates for the values of the coefficients. It was first formulated in 1935 by German physicist Carl Friedrich von Weizsäcker, and although refinements have been made to the coefficients over the years, the structure of the formula remains the same today.
The SEMF gives a good approximation for atomic masses and several other effects, but does not explain the appearance of magic numbers of protons and neutrons, and the extra binding-energy and measure of stability that are associated with these numbers of nucleons.
10 ) Nuclear Shell Model
Maria Goeppert Mayer, who made important discoveries about nuclear structure, is one of only two women to have won the Nobel Prize in physics.
In August 1948, Goeppert Mayer published her first paper detailing the evidence for the nuclear shell model, which accounts for many properties of atomic nuclei.
During her time at Chicago and Argonne in the late 1940s, Goeppert Mayer developed a mathematical model for the structure of nuclear shells, which she published in 1950. Her model explained why certain numbers of nucleons in an atomic nucleus result in particularly stable configurations. These numbers are what Eugene Wigner called magic numbers: 2, 8, 20, 28, 50, 82, and 126. Enrico Fermi provided a critical insight by asking her: "Is there any indication of spin orbit coupling?" She realised that this was indeed the case, and postulated that the nucleus is a series of closed shells and pairs of neutrons and protons tend to couple together. She described the idea as follows:
Think of a room full of waltzers. Suppose they go round the room in circles, each circle enclosed within another. Then imagine that in each circle, you can fit twice as many dancers by having one pair go clockwise and another pair go counterclockwise. Then add one more variation; all the dancers are spinning twirling round and round like tops as they circle the room, each pair both twirling and circling. But only some of those that go counterclockwise are twirling counterclockwise. The others are twirling clockwise while circling counterclockwise. The same is true of those that are dancing around clockwise: some twirl clockwise, others twirl counterclockwise.
Three German scientists, Otto Haxel, J. Hans D. Jensen, and Hans Suess, were also working on solving the same problem, and arrived at the same conclusion independently. Their results were announced in the issue of the Physical Review before Goeppert Mayer 's announcement in June 1949. Afterwards, she collaborated with them. Hans Jensen co-authored a book with Goeppert Mayer in 1950 titled Elementary Theory of Nuclear Shell Structure. In 1963, Goeppert Mayer, Jensen, and Wigner shared the Nobel Prize for Physics "for their discoveries concerning nuclear shell structure." She was the second and most recent female Nobel laureate in physics, after Marie Curie.
11 ) Ionic character percentage can be calculated by an Empirical formula if electronegativity values are given