How Molecular weight is Measured ?
Weight of an unknown compound can be determined by Physical,
Chemical and by Instrumental methods.
Physical methods include
1. Victor Meyer’s method
2. Molecular weight from elevation in boiling point
3. Molecular weight from depression in freezing point
4. Molecular weight from Osmotic pressure
5. Molecular weight from relative lowering of vapour pressure
6. Molecular weight by Dumas Method
Chemical Methods
7. Volumetric Methods
8. Silver salt method
9. Chloroplatinate method
Instrumental method
10. Mass spectrometry
In addition to all these methods weight of polymers can be determined by the methods like Gel Permeation Chromatography (GPC), Viscosity Methods etc.
___________________________________________________________________________________________________
Physical Methods
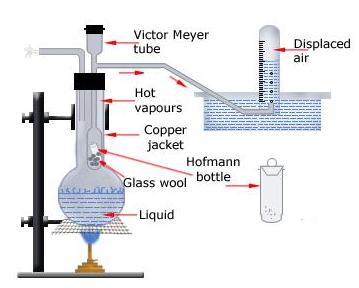
1. Victor Meyer's Method
Victor Meyer's method is used for the determination of molecular masses of volatile substances.
Principle
One mole of any volatile substance when vapourized gives vapour that occupy 22.4 L of volume under NTP conditions.
Apparatus
The apparatus used in this method consists of the following components:
Victor Meyer's tube
This is a hard glass tube of 1 cm2 cross-sectional area of 75 cm in length. The tube has a bulb at its lower end and an exit tube at the upper end of the tube.
Outer copper jacket
The outer copper jacket has a round bulb at its lower end. The victory Meyer's tube is suspended into this jacket with the help of a hard rubber cork.
Hoffmann's bottle
This is a small (3 mm diameter, 35 mm length) glass bottle with a very light stopper. This is used for introducing the sample into the Victor Meyer's tube.
Graduated glass tube
This is a burette-like glass tube graduated in milli litres. This is used for collecting the displaced air.
Process
A liquid (water), having boiling point about 20° to 30°C higher than that of the given liquid (whose molecular mass is to be determined) is poured into the outer jacket so as to fill its bulb up to one-third of its capacity. Cleaned and dried Victor Meyer's tube is suspended into the outer copper jacket, so that its lower end is just above the liquid in the outer jacket. The liquid in the outer jacket is heated to boiling and the air expelled from inside the Victor Meyer's tube is allowed to escape through the exit tube. When bubbling in the water stops, a water-filled graduated glass tube is placed over the end of the exit tube.
The given liquid compound (about 0.2 to 0.3 g) is taken in the Hoffmann's tube and weighed accurately. This tube is slipped into the Victor Meyer's tube and the cork replaced as quickly as possible. The liquid inside the Hoffmann's tube gets vapourised and the vapour so formed displaces equal volume of air out of the Victor Meyer's tube. This displaced air is collected in the graduated glass tube.
The graduated tube is carefully removed from the trough to a jar filled with water when the volume of the displaced air inside the graduated tube becomes constant. The volume of the air displaced is measured by equalizing the level of water inside and outside the tube. Temperature of the water in the jar, and the atmospheric pressure are also recorded.
Physical methods include
1. Victor Meyer’s method
2. Molecular weight from elevation in boiling point
3. Molecular weight from depression in freezing point
4. Molecular weight from Osmotic pressure
5. Molecular weight from relative lowering of vapour pressure
6. Molecular weight by Dumas Method
Chemical Methods
7. Volumetric Methods
8. Silver salt method
9. Chloroplatinate method
Instrumental method
10. Mass spectrometry
In addition to all these methods weight of polymers can be determined by the methods like Gel Permeation Chromatography (GPC), Viscosity Methods etc.
___________________________________________________________________________________________________
Physical Methods
1. Victor Meyer's Method
Victor Meyer's method is used for the determination of molecular masses of volatile substances.
Principle
One mole of any volatile substance when vapourized gives vapour that occupy 22.4 L of volume under NTP conditions.
Apparatus
The apparatus used in this method consists of the following components:
Victor Meyer's tube
This is a hard glass tube of 1 cm2 cross-sectional area of 75 cm in length. The tube has a bulb at its lower end and an exit tube at the upper end of the tube.
Outer copper jacket
The outer copper jacket has a round bulb at its lower end. The victory Meyer's tube is suspended into this jacket with the help of a hard rubber cork.
Hoffmann's bottle
This is a small (3 mm diameter, 35 mm length) glass bottle with a very light stopper. This is used for introducing the sample into the Victor Meyer's tube.
Graduated glass tube
This is a burette-like glass tube graduated in milli litres. This is used for collecting the displaced air.
Process
A liquid (water), having boiling point about 20° to 30°C higher than that of the given liquid (whose molecular mass is to be determined) is poured into the outer jacket so as to fill its bulb up to one-third of its capacity. Cleaned and dried Victor Meyer's tube is suspended into the outer copper jacket, so that its lower end is just above the liquid in the outer jacket. The liquid in the outer jacket is heated to boiling and the air expelled from inside the Victor Meyer's tube is allowed to escape through the exit tube. When bubbling in the water stops, a water-filled graduated glass tube is placed over the end of the exit tube.
The given liquid compound (about 0.2 to 0.3 g) is taken in the Hoffmann's tube and weighed accurately. This tube is slipped into the Victor Meyer's tube and the cork replaced as quickly as possible. The liquid inside the Hoffmann's tube gets vapourised and the vapour so formed displaces equal volume of air out of the Victor Meyer's tube. This displaced air is collected in the graduated glass tube.
The graduated tube is carefully removed from the trough to a jar filled with water when the volume of the displaced air inside the graduated tube becomes constant. The volume of the air displaced is measured by equalizing the level of water inside and outside the tube. Temperature of the water in the jar, and the atmospheric pressure are also recorded.
Calculations
Let,
The mass of the organic compound taken be = W g
Volume of air displaced = V1 mL
Atmospheric pressure = P mm of Hg
Room temperature, T1 = toC = (t + 273)K
Aqueous tension at toC = p mm of Hg
Pressure of the dry air, P1 = (P-p) mm Hg
The volume of the displaced air is equal to the volume of vapours formed from the given mass of the compound. So, by using the mole concept, one can write,
V mL of vapour weigh = W g
Therefore,
Problem
A volatile organic compound weighing 0.2 g, on heating in Victor Meyer's tube, displaced 30 mL of air at 27oC and 756 mm atmospheric pressure. Determine the molecular mass of the compound (Aqueous tension at 27C = 26 mm).
Solution
Mass of the substance taken = 0.2 g
Volume of air displaced = 30 mL
Temperature = 27oC = (27 + 273) K = 300 K
Atmospheric pressure = (756 - 26) mm Hg = 730 mm Hg
Then,
Therefore,
Volume of the vapours at NTP = 22.5 mL
Then,
2. Molecular Weight by Boiling Point Elevation
Background
This experiment demonstrates the use of colligative properties. The goal is to measure the molecular weight of a non-volatile solute by determining the concentration dependence of the boiling point elevation of a solution. The solvent used must be one of the compounds commonly referred to as volatile; that is, it must have an appreciable vapour pressure. One of the several useful aspects of colligative properties is the fact that the vapour pressure of volatile solvents is lowered when a non-volatile solute is used to make a solution. The result is that such a solution will necessarily have a higher boiling point than that of the pure solvent. The higher boiling point is due to the fact that a higher temperature is needed in the presence of the non-volatile solute, which is not making any contribution to the solution’s vapour pressure, in order to cause the volatile component of the solution, the solvent, to exert one atmosphere of pressure. It must be remembered that the boiling point elevation being investigated in this experiment is a property of the solution as a whole and, for ideal dilute solutions, is directly proportional to the solute concentration as shown in Equation 1.
∆Tb = m • Kb 1
In Equation 1, m is the solution molality and Kb is the boiling point elevation constant which is a function of the solvent not the solute. The value of ∆Tb is the boiling point of the solution minus that of the pure solvent, Tb*
. Based on the definition of molality it is possible to rearrange Equation 1 into Equation 2 in order to isolate the solute molecular weight, M.
M = [g • Kb] / [G • ∆Tb] 2
In Equation 2, ∆Tb is the boiling point elevation of a solution containing g grams of solute in G kilograms of solvent. It is interesting to note that Kb for a solvent may be estimated by Equation 3.
Kb = [R • Tb*2] / [∆Hvap ] 3
The value of Tb* in Equation 3 is, as mentioned, that of the boiling point of the pure solvent while ∆Hvap is the heat of vapourization of the pure solvent. Frequently, for dilute ideal solutions, the boiling points of the pure solvent and that of the solution may be used interchangeably.
Equations 1, 2, and 3 assume non-volatile and non-electrolyte solutes in ideal dilute solutions. Since colligative properties are independent of the identity of the solute, depending only on total particle concentration, it is possible to obtain important information about electrolytes by measuring boiling point elevations. This is especially true in the case of weak electrolytes that re only partially dissociated in solution. To the extent that a solute dissociates in solution the net number of actual particles present will increase. A larger number of particles in solution will result in a larger measured boiling point elevation, ∆Tb, because the molality, m, in Equation 1 will be larger. In the case of a weak electrolyte it is more appropriate to re-write Equation 1 as shown in Equation 4.
∆Tb = Kb • mapp = Kb • [g / (G • Mapp)] 4
In equation 4, mapp is the “apparent” total particle molality that results from the partial dissociation of the solute. Mapp is a weighted average molecular weight representative of the actual ions and molecules as they exist in solution.
The value of mapp may be evaluated in terms of the analytical molality, which is based on formula weight of solute, and the percent of the solute particles which undergo dissociation, @, as shown in
Equation 5.
mapp = n • @ • mo + (1 - @) • mo 5
In equation 5, n represents the number of ions produced by the dissociation of one molecule of solute while mo represents the molality based on the formula weight of the solute ignoring any dissociation. Equation 5 can be rearranged into equation 6 where g and G have the same meaning they did in equation 2 while Mo is the actual formula weight of the solute.
mapp = [{n - 1} • @ + 1] • [g / (G • Mo)] 6
The value of mapp may be evaluated as shown in Equation 7 with g, G, and Mapp as previously defined.
mapp = g / [G • Mapp] 7
Equations 6 and 7 may be combined to show that
@ = [Mo - Mapp ] / [Mapp • { n - 1}] 8
Since Mapp may be evaluated from the boiling point elevation as shown in Equation 4 it is easy
to determine the degree of dissociation of a weak electrolyte using Equation 8.
Inter-particle Forces
In addition to the influence of electrolyte behaviour the boiling point of solutions may be affected by the way in which solute particles interact with one another. Typically attractive forces exist between solute particles. These forces can have an influence on the value of the molecular weight as calculated using Equation 2. Equation 2 suggests a possible approach to attempt to correct for such inter-particle forces. A plot can be constructed of calculated molecular weight on the y-axis versus solution molality on the x-axis. Such a plot is extrapolated to zero molality, the y-intercept, to obtain what is referred to as the limiting molecular weight. This infinite dilution extrapolated molecular weight value may be thought of as the value of the molecular weight that would be obtained if only a single molecule were present in solution. Such a technique has the result of minimizing the influence of inter-particle forces.
Since Equation 2 requires values for the Kb constants, the values for common solvents are shown
in Table 1. Notice that since Kb is sensitive to atmospheric pressure, the information is provided in Table 1 so that Kb values may be corrected to the barometric pressure in the laboratory at the time the experiment is conducted.
A final note of caution is appropriate. The heart of this experiment is the measurement of how the boiling point of a pure solvent changes as a solution is prepared. The only reliable manner to accomplish this measure of temperature change is to make an actual measurement of the boiling point of the pure solvent at the actual laboratory conditions rather than depending on literature values. Since boiling points show significant pressure variation the boiling point of the pure solvent may be significantly different from the literature value if the barometric pressure in the laboratory is significantly different from standard atmospheric conditions. Failure to actually measure the boiling point of the pure solvent under the conditions that the solution boiling points are measured can give rise to serious errors!
Procedure
Apparatus and Materials Needed:
1 Cottrell boiling-point apparatus or equivalent (standard reflux/distillation set-up may be
used if Cottrell apparatus not available; ask instructor for assistance). 4
2 Digital thermometer capable of measuring to 0.1 ˚C equipped with a vapour phase
temperature probe (a Beckmann thermometer is better if available).
3 Suitable solvent and solute as suggested by instructor (benzoic acid, urea, oxalic acid, or
naphthalene as the solute and acetone, methanol, or ethanol as the solvent).
Boiling Point Measurement
If a Cottrell-type boiling point apparatus is available it will include suitable means for withdrawal of solution aliquots. When a Cottrell apparatus is unavailable it may be necessary to use a reflux/distillation set-up. When using a reflux set-up it is advisable to secure a piece of clean cotton cloth to the bulb of the thermometer. The cotton cloth, as it becomes wetted with condensing vapour, will insure the thermometer bulb is actually in contact with liquid rather than vapour. If a reflex/distillation set-up is used be sure that a two-neck round bottom flask is used in order to facilitate the addition of solute and removal of solution aliquots. A convenient size to use is a 250 ml round bottom flask. The round bottom flask is fitted with a reflex column mounted vertically in one of the necks. The second neck has a stopper inserted. At the top of the reflux column an adapter is fitted to permit the positioning of the thermometer as well as to permit the attachment of a guard condenser column to avoid escape into the laboratory of vapour as the solutions are boiled. Water cooling should be used as appropriate, particularly in the guard column.
Begin by placing about 250 ml (graduated cylinder) of the solvent into the round bottom flask and assembling the apparatus. Using a safe heat source as directed by the instructor heat the pure solvent to establish a stable reflux situation. Allow sufficient time for a stable temperature indication and record this value as the boiling point of the pure solvent under the current laboratory barometric pressure. Remove the heat source long enough for the boiling to subside then carefully introduce, through the second neck, enough solid solute to produce a solution whose weight percent concentration is 2% to 5%. For some solids, for example benzoic acid, it may be advisable to prepare solid pellets using a pellet press. Your instructor will provide assistance with this if it is necessary. After introduction of solute the boiling point of the solution is measured in the same way in which the boiling point of the pure solvent was measured. Be sure to allow sufficient time for a stable boiling point to be reached.
After the boiling point of the solution has been recorded remove the heat source and allow the boiling to subside. At this point a pipet is used to withdraw a sample of solution from the flask through the second neck. It is convenient to remove approximately 10 ml of sample although it is not necessary to accurately measure the actual volume nor that all aliquots are of the same volume. Each aliquot should be placed into a securely sealed weighing bottle and placed in a cold bath to promote rapid cooling. The empty weighing bottle should be previously weighed on an analytical balance. For efficient use of time the weighing bottle and its contents can be set aside as more solution samples are collected. Once several samples have been collected all of the solutions may be analyzed to determine the solution molality. Each subsequent solution is prepared from the previous one by adding, through the second neck of the round bottom flask, enough solute to produce a boiling point increase of about 0.1 to 0.2 degrees.
Each solution’s boiling point is measured and recorded. An aliquot of each solution is withdrawn and placed in a separate pre-weighed weighing bottle that is then cooled and set aside. Collect aliquots from four or five different solutions as directed by the instructor. Each of these aliquots are analysed as described below.
Once its contents have been cooled to room temperature each bottle and its contents are weighed
on an analytical balance. These data permit the weight of solution to be determined. The top is
now removed from the weighing bottle and the solvent is evapourated off. Typically a hot water
bath may be used to accomplish the solvent evapouration. If directed by your instructor a drying
oven may be used to remove the final residual of solvent to leave only solid solute in the
weighing bottle. Cool the weighing bottle and the solid solute to room temperature and weigh it.
Determine the weight of the solid solute (g in Equation 2). The weight of the solvent (G in
Equation 2) may be determined by the difference.
Calculations
Equation 2 may be used to calculate the molecular weight of the solute for each of the different solution concentrations. Use g and G for each of the solutions to calculate the molality of the solution. Prepare a graph of molecular weight, M, on the y-axis versus the solution molality, m, on the x-axis. The resultant line may be extrapolated to zero molality to obtain what is referred to as the “limiting molecular weight, Minfinite”. Calculate the degree of dissociation of the solute for each of the solutions whose boiling points have been measured. Prepare a plot of the degree of dissociation, @, on the y-axis versus the molality, mo, on the x-axis and extrapolate to zero concentration to obtain a limit at infinite dilution of the degree of dissociation, @infinite.
Problems:
Example 1: 0.64 g of adrenaline in 36.0 g of CCl4 produces a bp elevation of 0.49 °C. What is adrenaline's molecular weight?
Solution
1) Determine number of moles of adrenaline in solution:
ΔT = i Kb m
0.49 °C = (1) (4.95 °C / m) (x / 0.0360 kg)
x = 0.0035636 mol
From here it is a simple step: grams divided by moles equals the desired answer.
0.64 g / 0.0035636 mol = 180 g/mol (to two sig figs)
Notice that the Kb value for CCl4 was not given in the problem. In a textbook, the value would be in a chapter table or in an appendix. By the way, the textbook value for adrenaline's molecular weight is 183.204 g/mol
Example 2: 2.60 grams of a compound know to contain only indium and chlorine is dissolved in 50.0 g of tin(IV) chloride (Kb = 9.43 °C kg mol¯1). The normal boilng point is raised from 114.1 °C for pure SnCl4 to 116.3 °C for the solution. What is the molecular weight and probable molecular formula for the solute?
Solution
1) Keep in mind that the units on molecular weight are grams per mole. We already have the grams, what we now need are the moles that 2.60 grams represent. We can get that by the molality (moles solute / kg solvent):
ΔT = i Kb m
2.2 = 9.42 (x / 0.050); x = 0.01168 mol
2) Now we calculate the molecular weight:
2.60 g / 0.01168 mol = 222.6 g/mol
The probable molecular formula is InCl3, but I will let you ponder on how I did it.
Example 3: 5.00 g of an organic solid is dissolved in 100.0 g of benzene. The boiling temperature of this solution is 82.42 °C. The organic compound is 15.72% nitrogen, 7.92% hydrogen, 35.92% oxygen and the remainder is carbon. The boiling temperature of pure benzene is 80.1 °C; Kb = 2.53 °C-kg/mol. a. Determine the molecular weight of the organic solid.
Solution:
1) Determine moles of solute using boiling point elevation data:
Δt = Kb m
1.42 °C = 2.53 °C-kg/mol (x / 0.100 kg)
1.42 °C = (25.3 °C/mol) (x)
x = 0.0561265 mol
2) Use moles and 5.00 g to determine molecular weight (answer to part a):
5.00 g / 0.0561265 mol = 89.0845 g/mol
89.1 g/mol (to 3 sig figs)
0.8989 g/mL = 105.0 g / x
3. Molecular Weight by Freezing Point Depression
Investigations into the nature of solutions have shown that certain properties are different for the solution compared to the pure solvent. Foremost among these is a lower vapour pressure for the solution compared to the pure solvent. As a result of this effect, the freezing point of a solution is lower than the pure solvent, its boiling point is higher, and the solution exhibits osmotic pressure. These properties are called the colligative properties. These depend only on the total concentration of all dissolved species in the solution and not on their molecular or ionic nature. Thus, if 1.00 mole of glucose (C6H12O6) is dissolved in 1000 g of water, it will exhibit the same colligative properties as 0.50 mole of NaCl since the NaCl dissociates into 0.50 mole of Na+ and 0.50 mole of Cl– for a total concentration of species equal to 1.00 mole in 1000 g of water.
In this experiment, the freezing point depression of an aqueous solution of a solute will be compared to the freezing point of pure water. The depression of the freezing point can be used to calculate the molecular weight of the solute. If ∆Tf represents the depression of the freezing point, then:
∆Tf = Tf˚ – Tf
where Tf˚ is the freezing point of pure water and Tf is the freezing point of the solution. The
value of Tf is related to the concentration by the equation:
∆Tf = Kf m
where m is the molality of the solution, equal to (moles solute)/(kg solvent) and Kf is the molal
freezing point depression constant which, for water, is 1.86 C/molal. Other solvents can also be
used and each will have its own value of Kf. Tables of Kf values can be found in most general chemistry textbooks or chemistry handbooks. Regardless of the solvent, however, the equation, ∆Tf = Kf m applies well only to dilute solutions.
The molecular weight of a solute can be calculated from the freezing point and molality data. This can be seen if the equation is rearranged, making the indicated substitutions:
∆Tf = Kf m where m = (moles solute)/(kg solvent)
Since (moles solute) equals (g solute)/(MW solute), then:
Thus, by first determining the freezing point of pure water, then by carefully weighing out both a sample of solute and a sample of solvent water and determining the freezing point of the solution, the molecular weight of the solute can be calculated.
Determination of Molecular weight
Purpose:
To determine the molecular weight of an unknown organic compound by measuring the freezing point depression of an aqueous solution of the compound.
Introduction
The freezing point of a solution of a nonvolatile solute is less than that of the pure solvent. The freezing point depression or change in freezing point is given by eq. 1 and 2.
∆Tf = Kfm (1)
∆Tf = Tf - Tf° (2)
Tf is the freezing point of the solution in °C and Tf° is the freezing point of the pure solvent, also in °C. Kf is the freezing point depression constant. Its value depends only on the solvent. For water, Kf = -1.86°C/molal, m is the molal concentration of the solute, given by equation 3.
m = moles solute (3)
kg solvent
m = (grams solute)/MW (4)
kg solvent
The molecular weight of the solute is MW. To determine the molecular weight of an unknown from the freezing point depression, the freezing points of the pure solvent and of the solution are measured, and ∆Tf calculated from eq. 2. The molality of the solution is then calculated from eq. (1). The molecular weight of the solute is obtained by rearranging eq. 4 to solve for MW.
MW = grams solute (5)
(m)(kg solvent)
In this experiment you will determine the molecular weight of an unknown substance by measuring the freezing point depression of an aqueous solution of the unknown.
Experimental Procedure
Part 1. Freezing point of pure water:
The apparatus to be used is shown in Figure 3. Clean and dry the smaller test tube, and add to it approximately 20 mL of deionized water. Insert the stirrer, stopper and thermometer into the tube. Make sure that the thermometer is immersed in the water, and that the stirrer moves up and down freely.
Figure 3. Apparatus for determining the freezing point of pure water or a solution.
Prepare an ice-salt bath using generous quantities of rock salt. Place a layer of crushed ice 3-4 cm deep at the bottom of a 600 mL beaker. Sprinkle a portion of the salt on the ice. Continue to alternate the layers of ice and salt until the beaker is full. Stir the ice-salt mixture well, immerse the smaller test tube that contains the water into the ice-salt bath, and allow the water to cool to about 4°C. Remove the test tube from the bath, place it inside the larger test tube, and immerse the entire test tube assembly into the ice-salt bath, as shown in Figure 3. Stir the water, measure its temperature, and continue taking temperature measurements every 30 seconds. Estimate the temperature to the nearest 0.1°C. Stir the water in the test tube continuously at a uniform rate with the wire stirrer. Continue taking temperature-time readings until 6 successive readings are equal to one another. Repeat this measurement once.
Part 2. Determining the freezing point of a solution of an unknown substance.
Use the same thermometer you used in Part 1. Obtain a sample of unknown compound. Be sure to record the number of your unknown in your notebook. Weigh out 3.5 g to 4 g of the unknown to the nearest 0.001g. All masses should be recorded directly in your notebook.
Dry the smaller test tube and weigh it to the nearest 0.001 g. Add 20 ml of deionized water and weigh again to the nearest 0.001 g. WARNING: you must stir continuously while adding the unknown to the water or it can clump. Add the weighed unknown to the test tube. Stir the solution until the solid compound is completely dissolved.
Reweigh the test tube and contents to the nearest 0.001 g. Insert the stopper, thermometer, and stirrer. Make certain that the thermometer bulb is immersed in the solution.Discard the ice-salt bath used in Part 1, and prepare a fresh one.
Immerse the test tube containing the solution of unknown in the bath and allow it to cool to about 1°C. Remove it from the bath, place it in the larger test tube and place the entire test tube assembly back into the bath, as shown in Figure 3. Repeat the procedure in Part 1. That is, take temperature time readings to the nearest 0.10C every 30 seconds. Stir the solution continuously at a uniform rate during the course of the determination. Continue taking readings until the solution solidifies. Do another determination with a second portion of your unknown.
Results
Calculations
1. For each trial, prepare a graph of time versus temperature as shown in Figures 1 and 2. Determine the freezing point from each graph.
2. Calculate the average value of the freezing point of pure water from the results of the two trials in Part 1.
3. Calculate the freezing point depression for each of the two trials in Part 2 (eq. 2)
4. For each trial in Part 2, calculate the mass of water and the mass of unknown compound.
5. Calculate the molality of each solution in Part 2 (eq. 1).
6. Calculate the molar mass of the unknown for each solution (eq. 3).
7. Calculate the average molar mass of your unknown.
Problems:
Example 1: Vitamin K is involved in the blood clotting mechanism. When 0.500 g is dissolved in 10.0 g of camphor, the freezing point is lowered by 4.43 °C. Calculate the molecular weight of vitamin K.
Solution
1) To solve this problem, I'd like to engage in an analysis of the units. We will start with the freezing point depression equation:
ΔT = i Kf m
Replacing the right side with units gives: ΔT = (°C kg mol¯1) times (mol kg¯1)
Notice that i goes away since it is unitless. Next I will replace mol with g / g mol¯1:
ΔT = (°C kg mol¯1) times (g / g mol¯1 kg¯1)
2) Now, let's insert numbers in the proper place:
4.43 = (40.) times (0.500 / x 0.010)
x is the molecular weight of Vitamin K, 0.500 and 4.43 are from the problem and 0.010 is 10 g of camphor done as kilograms. This becomes:
4.43 = 2000 / x
x = 8860 g / mol
We know this is a reasonable answer since vitamins and proteins have molecular weights in the thousands or even tens of thousands.
Example 2: A compound containing only boron, nitrogen, and hydrogen was found to be 40.3% B, 52.2% N, and 7.5% H by mass. When 3.301 g of this compound is dissolved in 50.00 g of benzene, the solution produced freezes at 1.30 °C. The freezing point of pure benzene is 5.48 °C; Kb for benzene is 5.12 °C m-1. What is the molecular weight of this compound?
a. Determine the molecular weight of the solid.
b. Determine the molecular formula of the solid
c. Determine the mole fraction of the solid in the solution
d. If the density of this solution is 0.8989 g/mL , calculate the molarity of the solution
Solution:
1) Determine moles of solute using freezing point depression data:
Δt = Kf m
4.18 °C = 5.12 °C-kg/mol (x / 0.0500 kg)
4.18 ° = (102.4 °C/mol) (x)
x = 0.04082 mol
2) Use moles and 3.301 g to determine molecular weight (answer to part a):
3.301 g / 0.04082 mol = 80.867 g/mol
80.9 g/mol (to 3 sig figs)
3) Use an on-line empirical formula calculator to determine the empirical formula:
BNH2
4) Determine the molecular formula (answer to part b):
the "empirical formula weight" of BNH2 is 26.8338 g
the molecular weight divided by the "EFW" yields 3
therefore, the molecular formula is:
B3N3H6
Comment: the particular substance in this question is Borazine, often referred to as the "inorganic benzene." Its formula is written (BH)3(NH)3.
5) Determine the mole fraction of the solute (answer to part c):
moles of solvent = 50.00 g / 78.1134 g/mol = 0.6401 mol
χsolute = 0.04082 mol / (0.04082 mol + 0.6401 mol)
χsolute = 0.060
6) Determine molarity of solution (answer to part d):
i) our solution weighs 53.301 g; calculate its volume:
0.8989 g/mL = 53.301 g / x
x = 59.30 mL
ii) calculate the molarity:
0.04082 mol / 0.05930 L = 0.6884 M (to 4 sig figs)
Example 3: A 0.2436 g sample of an unknown substance was dissovled in 20.0 mL of cyclohexane. The density of cyclohexane is 0.779 g/mL. The freezing point depression was 2.50 °C. Calculate molar mass of the unknown substance.
Solution:
1) Determine grams of cyclohexane:
(0.779 g/mL) (20.0 mL) = 15.58 g
this is 0.01558 kg
2) Determine moles of unknown substance present:
Δt = i Kf m
2.50 °C = (1) (20.2) (x / 0.01558)
x = 0.00192822 mol
Note assumption of i = 1 for the solute. Also, the cryoscopic constant for cyclohexane needed to be looked up.
3) Determine molecular weight:
0.2436 g / 0.00192822 mol = 126.3 g/mol
4. Determination of Molecular Mass from Osmotic Pressure
In order to determine the molecular mass of an unknown, non volatile compound, a known mass (say w g) of the compound is dissolved to prepare a known volume (say v liters) of solution. The osmotic pressure of the solution is determined and the molar mass is calculated as follows.
π = nBRT/v
where nB is the number of moles of the solute and is given by WB/MB.
Hence WB is the mass of the solute in gm and MB is the molecular mass of the solute.
Thus,
π = WBRT/MBV
MB = WBRT/πV
This method is exceptionally suitable for the determination of molecular masses of macromolecules such as proteins and polymers. This is because, for these substances the values of other colligative properties such as elevation in boiling point or depression in freezing point are too small to be measured. On the other hand, osmotic pressure of such substances are measurable.
5. Molecular mass from relative lowering of vapour pressure
When a non-volatile solute is added to a solvent, the vapour pressure is lowered due to the following reasons:
(i) Percentage surface area occupied by the solvent decreases. Thus the rate of evapouration and vapour pressure decreases. The solute molecules occupy the surface, and so the per cent surface area occupied by the solvent decreases.
(ii) According to Graham’s law of evapouration,
Rate of evapouration ∝ 1/√density
When a non-volatile solute is dissolved in a liquid, its density increases. Thus both rate of evapouration and vapour pressure are lowered.
If p0 is the vapour pressure of pure solvent and ps is the vapour pressure of the solution, the difference (p0 – ps) is termed lowering in vapour pressure and the ratio [p0–ps/p0] is termed relative lowering in vapour pressure.
Raoult, in 1886, established a relationship between relative lowering in vapour pressure and composition of the solution after a series of experiments in various solvents. The relationship is known as Raoult’s law. It states that the relative lowering in vapour pressure of a dilute solution is equal to mole fraction of the solute present in the solution.
If n moles of solute be dissolved in N moles of the solvent, the mole fraction of the solute will be n/n+N.
According to Raoult’s law, [p0–ps/p0] = n/n+N.
This is the mathematical expression for Raoult’s law.
(Modified form of Raoult’s law: The above relationship can be written as,
p0/p0–ps = n+N/n = 1 + N/n
or p0/p0–ps – 1 = N/n or p0/p0–ps = N/n
or p0–ps/ps = n/N = wA/mA × mB/wB
This equation gives accurate results and easy to apply.]
Derivation of Raoult’s law for a dilute solution
When a non-volatile solute is dissolved in a volatile solvent, a fraction of the surface of solvent is blocked by solute molecules where no evapouration occurs. Thus, under similar conditions, the vapour pressure is decreased. The vapour pressure of the solution, thus, depends upon the number of molecules of the solvent present on the surface of the solution.
The number of such molecules is proportional to mole fraction of the solvent.
So, the vapour pressure of solution,
‘ps’ ∝ N/n+N
or ps = k.N/n+N ….(i)
(k is proportionality factor)
For pure solvent, n = 0
And hence, p0 = k N/0+N = k …(ii)
Putting the value of k in Eq. (i)
or ps = p0 N/n+N
or ps/p0 = N/n+N
or 1 – ps/p0 = 1 – N/n+N
or p0–ps/p0 = n/n+N …(iii)
This is Raoult’s equation.
If a solution is made by dissolving wA g of solute (molecular mass mA) in wB g of the solvent (molecular mass mB), the mole fraction of the solute will be = wA/mA/wA/mA + wB/mB
If the solution is very dilute, wA/mA can be neglected in the denominator as compared to wB/mB. The Eq. (iii), thus, becomes
p0–ps/p0 = wA/mA × wB/mB …(iv)
This relationship is useful in the determination of the molecular mass of dissolved solute by measuring relative lowering of vapour pressure.
6. Determination of Molar Mass via the Dumas Method
1. Purpose
In this experiment you will determine the molar mass of an unknown liquid using a simplified variation of the Dumas method.
2. Background
In the early 19th century, Jean-Baptiste Dumas, a distinguished French chemist, created a relatively simple method for determination of the molecular weight of a substance. With this method, molecular weight is calculated by measuring the mass of a known volume of a vapourized liquid. Because the concept of the mole had not been developed in Dumas’ era, he computed relative molecular weights based on relative gas densities. Though Dumas got mixed results based on erroneous assumptions concerning elements in the gas phase, he is credited with establishing values for the molecular weights of thirty elements.
In the modern version of the Dumas procedure, an Erlenmeyer flask is used rather than the glass bulb of Dumas’ day. The temperature, pressure and volume of the vapour are determined and the molar mass is found utilizing the Ideal Gas Law.
3. Procedure
Overview: A sample of a volatile liquid is added to a pre-weighed flask. The flask is submerged in a boiling water bath to vapourize the liquid. Because an excess amount of liquid is used, the volume of vapour produced is greater than the volume of the flask. Upon heating, the vapour that is created initially pushes the air out of the flask and then the vapour begins exiting the flask until the pressure inside the flask is equal to the atmospheric pressure. The mass of the vapour remaining in the flask is obtained by reweighing the flask. Additional measurements are made to determine the pressure, temperature and volume of the sample. Three trials will be completed to determine the average molar mass of the unknown liquid. NOTE: All data should be tabulated. Data tables are to be prepared before attending lab.
A. Experimental apparatus
Assemble the apparatus as shown in figure 1. Be sure there is sufficient water in the 800 mL beaker to surround the bottom of the flask, but not so much that it overflows upon immersion of the flask. You do not need the foil and wire (or band) attached at this time. NOTE: To help assist observation of the unknown in part B, it is a good idea to mount the flask at a slight angle instead of perfectly vertically.
B. Vapourizing the Unknown
Remove the cap and add approximately 2 mL of unknown to the flask. Secure the cap to the flask, and immerse the flask in the heating bath. Secure the flask with the clamp. IT IS EXTREMELY IMPORTANT THAT NO WATER GETS INSIDE THE FLASK. Mount the temperature probe in the bath, so it is as close to the unknown as possible. As the bath heats the liquid and the liquid begins to boil, follow the vapour up through the flask with your eye. Soon vapour will start to escape through the pin hole. The vapour is easiest to see if you are at eye level with the top of the flask and look across the top of the flask. The vapour will look like a colourless swirling cloud or jet. (It also helps to look toward a light source.) When all of the liquid has vapourized, including any droplets that may have formed on the neck, no more vapour will be observed escaping through the pin hole (this should take no more than one to two minutes). NOTE: It can be difficult to observe the unknown vapourizing and streaming due to bubbling on the flask and water vapour generated by the bath. If seeing the vapourization is too difficult, simply allow the liquid to vapourize for approximately one minute after the bath has reached a steady boil. As soon as the vapourization is complete (no more liquid in the flask, no more visible vapour escaping through the pinhole), record the temperature of the bath and remove the flask from the bath. CAREFULLY, THOROUGHLY AND QUICKLY, dry the outside of the flask. IT IS EXTREMELY IMPORTANT THAT THE EXTERIOR OF THE FLASK BE COMPLETELY DRY. Allow the flask to cool to room temperature, weigh and record the mass. (Do not remove the cap before weighing.) Repeat section B two more times. It is not necessary to empty or clean the flask between trials. If necessary, add more water to the water bath for trials 2 and 3. When the heating bath is no longer needed, turn off the hot plate.
C. Obtaining the Volume and Pressure of the Vapour
After the final weighing, remove the cap and wire. Fill the flask completely full with water. Measure the amount of water the flask can hold using a graduated cylinder Record the barometric pressure. This is the same as the pressure in the flask.
D. Clean Up and Handling of Excess Unknown
Perform clean up and handling of excess unknown after the three trials in accordance with general laboratory guidelines and/or specific TA/Staff instructions.
4. Calculations
Before leaving lab,
a. Calculate the mass of unknown vapour in the chamber for each trial.
b. For each trial, calculate the number of moles of unknown vapour in the flask just after vapourization using the ideal gas law.
c. For each trial, calculate the molar mass of the unknown using n = mass/MM.
d. Calculate the mean value and standard deviation for the molar mass of the unknown.
e. Show your work to your instructor. If the calculation is correct, he/she will initial it and give you the name of your unknown. From its formula, calculate the molecular weight of the unknown.
f. Calculate the percent error of your measured molecular weight.
% error=|accepted value−experimental value |/accepted value ×100
5. Discussion/Conclusion
Report the mean value of the unknown molar mass and the % error. Compare your value to the accepted value of the molar mass. Consider the following when discussing your results: What assumptions are made in this procedure, how do they contribute to the error and how do the different sources of error impact the results? For example, here are some questions to get you started:
From the time the mass of the flask is first measured until the final measurement, it is handled a number of times with oily fingers. How does this technique affect the reported mass of the vapour in the flask, and the final calculated molar mass? Is this error significant relative to other sources of error?
What is the measurement error of the graduated cylinder? Is this significant relative to other sources of error?
The flask is completely filled with vapour only when it is removed from the hot water bath. However, when the flask cools some of the vapour condenses in the flask. As a result of this observation, will the reported molar mass of the liquid be too high, too low, or unaffected?
Problems:
How many grams of ethanol would occupy 750 mL at 790.5 torr pressure at 100oC? (molar mass of ethanol is 46.0 g mol-1)
Relavent equations:
PV = nRT
1 atm = 101.3 kPa
R = 0.8206 L atm mol-1 K-1
P = (750 torr)(1atm/760 torr) = 0.987 atm
V = 0.750 L
T = 373 K
You can calculate the moles of the gas if you know can measure the volume of gas:
(0.987 atm)(0.750 L) = n (0.8206 L atm mol-1 K-1)(373 K)
n = 0.0242 mol
And you can calculate the grams of gas if you know the molar mass:
(0.0242 mol)(1 mol / 46.0 g) = mass
mass = 1.11 g
Calculate the molar mass of a compound in the Dumas method for which a volume of the expermental container was 452 mL and the pressure was 745.1 torr. The difference in mass between the empty container and the final measurement was 1.129 g.
P = (745.1 torr)/(760 torr atm-1) = 0.9804 atm
V = 0.452 L
T = 373 K
(0.9804 atm)(0.452 L) = n (0.8206 L atm mol-1 K-1)(373 K)
n = 0.01448 mol
Molar mass = M, m = mass, n = moles
M = m/n
M = (1.129 g)/(0.01448 mol)
M = 78.0 g /mol
Chemical Methods:
7. Volumetric method Volumetric method is used for determining the molecular masses of acid and bases.
In this method, a known mass of the acid is dissolved in water or neutral alcohol, and the solution is titrated against a standard solution of an alkali using phenolphthalein as the indicator. From the volume of alkali required for neutralization of the acid, molecular mass can be calculated.
Calculations Let,
The mass of the acid dissolved be = W g
Volume of alkali used for complete neutralization of acid solution = V1 mL
Normality of the alkali solution = N1
Then, we can write,
V1 mL of alkali solution having normality N1 = Wg acid
From the definition,
1000 mL of 1N alkali solution = 1 gram equivalent of the alkali.
Since, acids and bases (or alkalies) neutralize each other in equivalent proportions, hence,
Therefore,
Then
Molecular mass of the acid = Equivalent mass of the acid x basicity of the acid
Problem About 0.000225 g of a monoacid base required 1 mL of 0.005 N H2SO4 for complete neutralization. Calculate the molecular mass of the base.
Solution For an acid-base reaction, at the neutralization point,
Number of equivalents of acid = Number of equivalents of base
=
Therefore,
Acidity of the base (given) = 1
Thus, Molecular mass of the base = 1 x 45 amu = 45 amu
8. Silver salt method Silver salt method is used for determining the molecular masses of carboxylic acids. This method is based on the fact that they form insoluble silver salts, which upon heating decompose to leave a residue of metallic silver.
Procedure
The unknown acid is dissolved in water and treated with a slight excess of ammonium hydroxide. The excess of ammonia is boiled off. To this sufficient quantity of silver nitrate is added when a white precipitate of silver salt is obtained. The precipitate is separated by filtration, washed successively with water, alcohol and ether and dried in the steam oven. About 0.2 g of the dry silver salt is weighed into a crucible and ignited until the decomposition is complete. Ignition is repeated until the crucible with the residue of silver has attained constant weight. The molecular mass of the acid is then calculated from the mass of the silver salt taken and the mass of the residue of metallic silver obtained from it.
For example, for a monocarboxylic acid, we can write
Calculations Let,
The mass of silver salt taken for ignition be = W g
Mass of silver left behind after ignition = W1 g
We have RCOOAg = Ag1 equivalent 1 equivalent
The equivalent mass of silver is 108. Hence, the mass of silver salt that would leave 108 g of residue (equivalent mass of the silver) is equal to the equivalent mass of the silver salt of the acid.
Then,
and,
Equivalent mass of acid = Equivalent mass of the silver salt - Equivalent mass of silver+ Equivalent mass of hydrogen
Or,
Then,
Molecular mass of the acid = Equivalent mass of the acid x Basicity of the acid.
The basicity of an acid is equal to the number of ionizable hydrogen atoms in a molecule of the acid.
Problem
About 0.759 g of silver salt of a dibasic acid was ignited when a residue of 0.463 g of metallic silver was left. Calculate the molecular mass of the acid.
Solution Mass of silver salt of carboxylic acid, W1= 0.759 g
Mass of silver left as residue, W = 0.463 g
= 177 - 108 + 1 = 70 amu
Molecular mass of acid = Equivalent mass x Basicity
= 70 x 2 amu = 140 amu
9. Chloroplatinate method
This method is used to find the molecular masses of organic bases.
Principle The given organic base is allowed to react with chloroplatinic acid (H2PtCl6) in concentrated HCl to form insoluble platinic chloride. This precipitate is separated, dried, weighed and is subsequently ignited till decomposition is complete. The residue left behind is platinum and is weighed again. The molecular mass is then calculated by knowing the mass of the platinic chloride salt and that of platinum left.
If B represents the molecule of monoacidic organic base, then the formula of platinum chloride salt is B2H2PtCl6.
Calculations Let the mass of platinic chloride salt be = W g
The mass of platinum residue left = x g
From the molar stoichiometry,
Number of mol of B2H2PtCl6 = Number of mol of Pt
Or
If E is the equivalent mass of the organic base,
Then molecular mass of the salt (B2H2PtCl6) = 2E + 2 + 195 + 213
= 2E + 410
Molecular mass of base = E x Acidity of the base
Problem
About 0.98 g of the chloroplatinate of some diacidic base when ignited left 0.39 g of platinum as residue. What is the molecular mass of the base? (Atomic mass of Pt = 198).
Solution
Mass of the chloroplatinate salt = 0.98 g
Mass of platinum = 0.39 g
Let E be the equivalent mass of the base, then
Molecular mass of salt (B2H2PtCl6) = 2E + 410
Molecular mass of salt = 40 x 2 = 80.
Instrumental Method:
10. Mass Spectroscopy
Mass spectroscopy is a modern technique that helps to measure the mass (molecular weight) of a molecule. In addition, one can also gain information on the structure of the organic compound.
In this method, a gaseous sample of the substance is bombarded with a high speed electron beam that has sufficient energy to do the following:
- Cause ionization of the molecules of the substance.
- Decompose the molecules into smaller positively charged fragments.
Fig:16.20 Schematic representation of an electronic impact, magnetic sector mass spectrometer
A small amount of the sample is introduced into the mass spectrometer and is bombarded by a stream of high-energy electrons (70 electron volts (eV) or 1600 kcal/mol). When the high-energy electron strikes an organic molecule, it removes a valence electron from the molecule, producing a cation radical (cation because the molecule has lost a negatively charged electron; radical because the molecule now has an odd number of electrons).
Electron bombardment transfers such a large amount of energy to the sample molecules that the cation radicals fragment. They fly apart into numerous smaller pieces, some of which retain a positive charge, and some of which are neutral. The fragments are then made to pass through a strong magnetic field applied perpendicularly, where they are deflected according to their mass-to-charge ratio (m/z).
Neutral fragments are not deflected by the magnetic field and are lost on the walls of the instrument. Positively charged fragments, however, are sorted by the mass spectrometer onto a detector, which records them as peaks. Since the number of charges 'z', is usually 1, the peaks represent the masses of the ions. The 'mass spectrum' of a compound is usually presented as a bar graph with 'unit' masses (m/z values) on the x-axis, and intensity (number of ions of a given m/z striking the detector) on the y-axis. The highest peak is called the 'base peak' and is arbitrarily assigned an intensity of 100%.
Fig:16.21 Mass spectra of methane, CH4 (molecular weight = 16)
The mass spectrum of methane is relatively simple, since few fragmentations are possible. The base peak has m/z = 16, which corresponds to the unfragmented methane cation radical or the 'molecular ion' (M+). The mass spectrum also shows ions at m/z = 15 and 14, corresponding to cleavage of the molecular ion into CH and CH fragments.
For larger molecules, the mass spectral fragmentation patterns are usually complex, and the molecular ion is often not the highest (base) peak.
Abnormal Molecular Masses
When the molecular mass of a substance as determined by using colligative properties doesnot come out to be same as expected theoretically, then it is said to show abnormal molecular mass.
Abnormal molecular mass is obtained when the substance in the solution undergoes dissociation or association.
Dissociation results in the increase in the number of particles hence increase in the value of colligative property in the value of colligative property and decrease in molecular mass as all the colligative properties are inversely proportional to molecular mass.
Association results in the decreases in the number of particles and hence decrease in the value of colligative property and increase in the molecular mass.
Electrolytes in water undergo dissociation, while in some non-polar solvents, two or more molecules of solute "associate to form bigger molecules, e.g. Acetic acid in benzene and benzoic acid in benzene undergo association to form dimers. This association is due to the formation of hydrogen bonding.
Van't Hoff factor:
Van't Hoff factor can be defined as the ratio of observed colligative property to normal or therotical colligative property. It is denoted by the letter 'i'. This factor was introduced to calculate the extent of
association or dissociation
.
The relation between degree of association and Vant Hoff factor
To calculate degree of dissociation, write the dissociation equation. Start with one
mole and suppose a is the degree of dissociation.
To calculate degree of association, write the association equation. Start with 1 mole
and suppose α is the degree of association,
To summarise