Davisson Germer Thomson Experiments showed that particles could act as waves
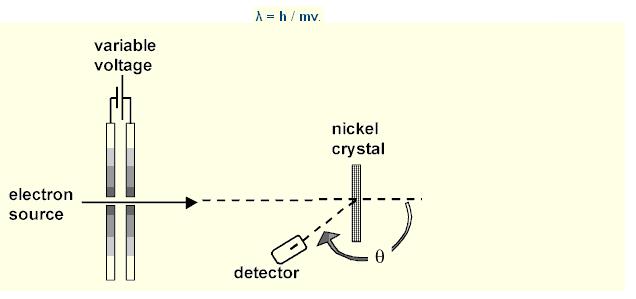
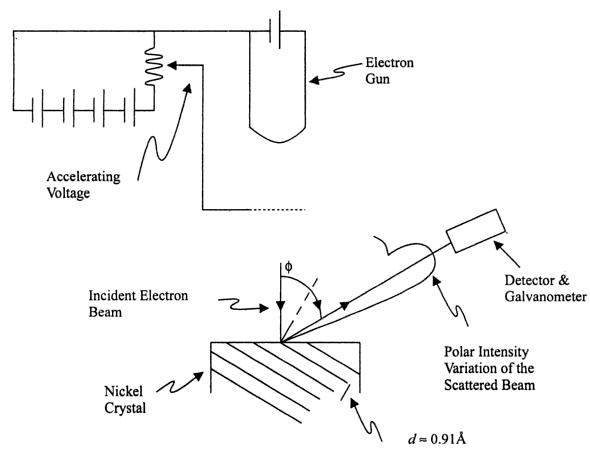
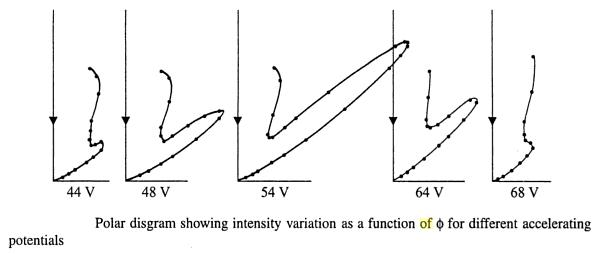
Three years after de Broglie asserted that particles of matter could possess wavelike properties, the diffraction of electrons from the surface of a solid crystal was experimentally observed by C. J. Davisson and L. H. Germer of the Bell Telephone Laboratory. In 1927 they reported their investigation of the angular distribution of electrons scattered from nickel. With careful analysis, they showed that the electron beam was scattered by the surface atoms on the nickel at the exact angles predicted for the diffraction of x-rays according to Bragg's formula, with a wavelength given by the de Broglie equation
Lambda = h / p or h / mv
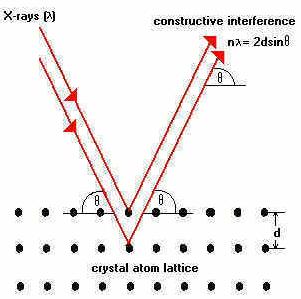
Sir William Lawrence Bragg, an X-ray crystallographer, had devised a method to calculate the positions of atoms within a crystalline material by observing the manner of diffraction of X-ray beam from the crystal lattice. X-rays have wavelengths of the order of the inter-atomic distance in crystals. 1 Angstrom = 10 to the power -10 meter. He discovered a fundamental relation for the determination of crystal structure called as “Bragg’s law of X-ray diffraction" (1912). Maxima of intensity (called as Bragg peaks) are formed where X-rays are reflected from crystal planes and satisfy the condition that the path-length difference of interfering X-rays is equal to an integer multiple of their wavelengths i.e. n ( Lambda ) = 2d Sin Theta
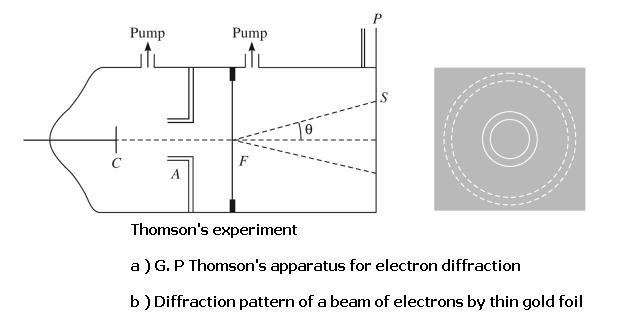
Also in 1927, G. P. Thomson, the son of J. J. Thomson, reported his experiments, in which a beam of energetic electrons was diffracted by a thin foil. Thomson found patterns that resembled the x-ray patterns made with powdered (polycrystalline) samples. This kind of diffraction, by many randomly oriented crystalline grains, produces rings. If the wavelength of the electrons is changed by changing their incident energy, the diameters of the diffraction rings change proportionally, as expected from Bragg's equation.
The diffraction patterns simulated above compare the effects of x-rays passing through a thin foil with those of high energy electrons passing through the same medium. Notice how similar the patterns are to each other when the de Broglie wavelength of an electron beam equals the wavelength of the original x-rays.
These experiments by Davisson and Germer and by Thomson proved that de Broglie's waves are not simply mathematical conveniences, but have observable physical effects. The 1937 Nobel Prize in Physics was awarded to these gentlemen for their pioneering work. Just as Compton showed that waves could act like particles, Davisson and Germer showed that particles could act as waves.
The diffraction rings had narrowly defined radii and always seemed to occur in multiples i.e. circles of radii 2r, 3r, ....
The radii of the different sets of rings were found to correspond precisely to the spacing of the various planes of atoms. Electrons were scattered at different angles from the atoms of crystallites and produced interference pattern with maxima corresponding to those angles satisfying the Bragg condition.
In terms of the probabilistic interpretation of matter waves, the probability of finding an electron scattered at an angle theta is exactly equal to computed intensity pattern of interfering waves associated with electron beam.
The radii of the different sets of rings were found to correspond precisely to the spacing of the various planes of atoms. Electrons were scattered at different angles from the atoms of crystallites and produced interference pattern with maxima corresponding to those angles satisfying the Bragg condition.
In terms of the probabilistic interpretation of matter waves, the probability of finding an electron scattered at an angle theta is exactly equal to computed intensity pattern of interfering waves associated with electron beam.
The radius of the rings decrease with increase in velocity and thus voltage.
So Radius of the rings are inversely proportional to root V