The Magic Numbers and "Doubly Magic " for Nuclear Isotope Stability
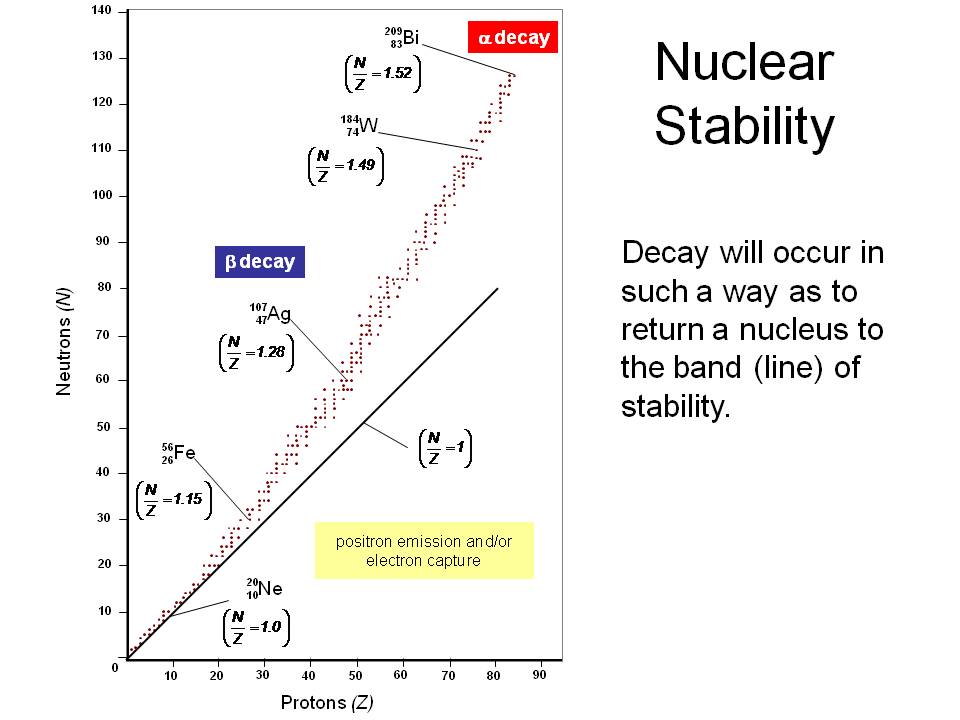
Nuclear Stability is a concept that helps to identify the stability of an isotope. The two main factors that determine nuclear stability are the
neutron/proton ratio and the total number of nucleons in the nucleus.
Although rare 4 Stable odd-odd nuclides exist 1H2 , 3Li6, 5B10, 7N14
neutron/proton ratio and the total number of nucleons in the nucleus.
Although rare 4 Stable odd-odd nuclides exist 1H2 , 3Li6, 5B10, 7N14
-
Before we discuss the details of Magic Numbers in Nucleus, let us recall the stability in electron configurations
-
The noble gases; helium, neon, argon, xenon and radon; are chemical inert; i.e., they are very stable chemically. The interpretation is that the electrons form shells and when a shell is filled the configuration is exceptionally stable and requires a lot of energy to knock an electron out of a filled shell. On the other hand, an electron in excess of a filled shell is very easy to remove from the atom. Thus the inertness of these elements is a consequence of the stability of the filled shells. The elements one electron beyond a filled shell are the highly reactive alkalai metals; lithium, sodium, potassium, cesium, rubidium and francium. The elements with one electron less than a filled shell are the highly reactive halogens; flourine, chlorine, bromine and iodine.
Hydrogen may also be consider a member of either one of these groups.
The atomic numbers of the noble gases are 2, 10, 18, 36, 54 and 86. These can be considered magic numbers for electron structure stability. The differences in these numbers are: 8, 8, 18, 18, 32. These differences are twice the value of the squares of integers; i.e., 2(2^2), 2(2^2), 2(3^2), 2(3^2), 2(4^2). The first number 2 in the series {2, 10, 18, 36, 54, 86} is also of the form of twice the square of an integer, 2(1^2).
The explanation of the magic numbers for electron structures is that there are shells for 2(n2) electrons where n=1, 2, 3, 4... The reason for the coefficient 2 in the formula is that there are two spin orientations of an electron. Pauli's exclusion principle operates and so electrons fill the states sequentially with no two electrons of an atom in the same state.
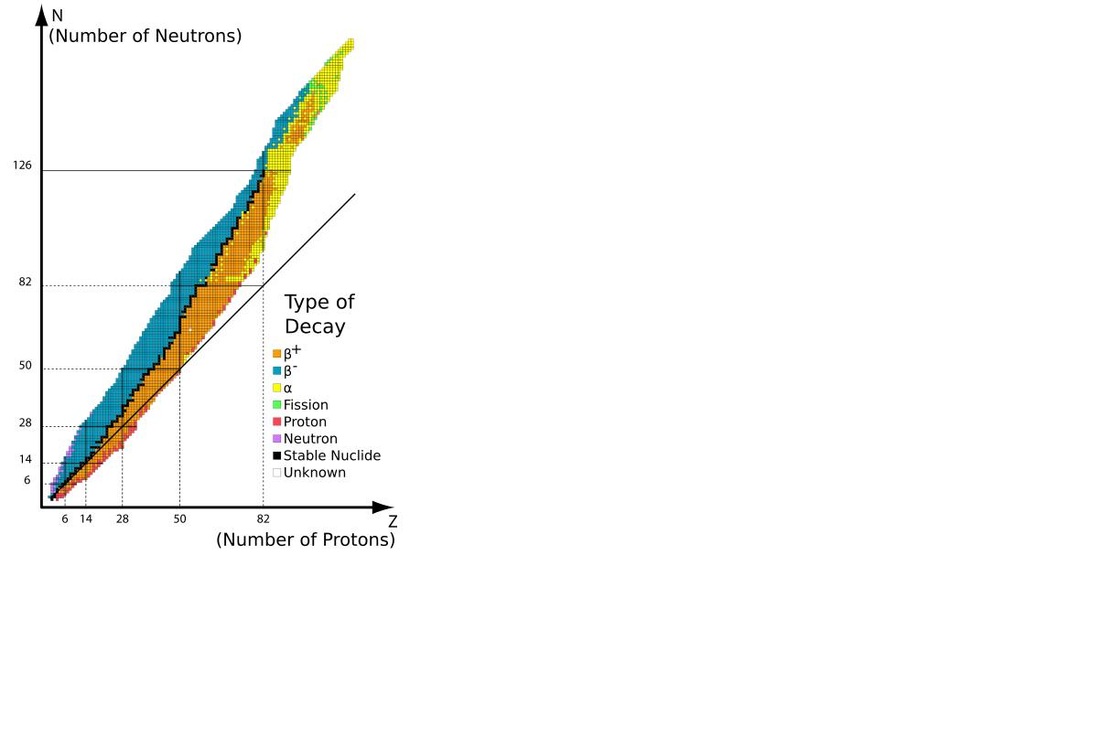
Nuclear Magic Numbers
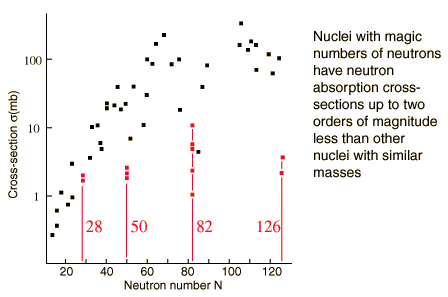
Maria Goeppert-Mayer and other physicists examining the properties of the isotopes of elements discerned that isotopes in which the proton and/or the neutron numbers were particular values have notable properties such as stability. These magic numbers are 2, 8, 20, 28, 50, 82, 126
It is profoundly significant that there are magic numbers for the proton and neutron numbers separately because that indicates that the protons and neutrons are organized in separate shells. In some respects a proton and a neutron appear to be simply different states of a basic particle called a neucleon.
However there apparently is a distinct differentiation in the nucleus of the the proton and neutron nucleons.
The magic numbers for atomic electron structure are perfectly explainable in terms of a formula. The increments in the particular stable atomic numbers are equal to twice the square of an integer. These increments correspond to the maximum occupancy levels of shells. Therefore in looking for an explanation of
the nuclear magic numbers it is reasonable to look at the increments in the magic numbers; i.e.,
Magic Numbers 2 8 20 28 50 82 126
Increments 2 6 12 8 22 32 44
The difference in consecutive magic numbers could be sums of shell occupancy levels. For example, the value of 44 might correspond to two shells of 22 each.
Likewise a value of 22 might correspond to two shells, one of occupancy 20 and one of occupancy 2.
The maximum occupancy levels for the atomic electron shells being the square of an integer n corresponds to the sum of the first n odd numbers. Let look at the sum of the first even numbers.
Even Numbers 2 4 6 8 10 12 14
Cumulative Sum 2 6 12 20 30 42 56
The cumulative sum sum values show up in the set of increments of nuclear magic numbers and the increments can be represented as simple sum of these cumulative sum values, as is shown below.
Increments of
Magic Numbers 2 6 12 8 22 32 44
Generation from
Cumulative Sums
of Even Numbers 2 6 12 2+6 2+20 2+30 2+42
If there is anything to this pattern the number 14=2+12 should be something like a magic number increment (rather than a magic number). This would mean that 22 and 34 should be nearly magic numbers. Titanium, atomic number 22, has five stable isotopes but scandium, the element with atomic number 21, has only one and vanadium, the element with atomic number 23 has only two. Selenium, atomic number 34, has six stable isotopes but bromine, the element with atomic number 35, has only two and arsenic, the element with atomic number 33 has only one. This is suggestive of the magic-ness of 22 and 34 and thus that 14=2+12 is a magic number increment.
These results look impressive but, as the table below shows, there is an even-odd alternation in the relationship between the number of stable isotopes and the atomic (proton) number.
Proton
Number 1 2 3 4 5 6 7 8 9 10
Number of
Stable Isotopes 2 2 2 1 2 2 2 3 1 3
Proton
Number 11 12 13 14 15 16 17 18 19 20
Number of
Stable Isotopes 1 3 1 3 1 4 2 3 2 5
Proton
Number 21 22 23 24 25 26 27 28 29 30
Number of
Stable Isotopes 1 5 1 3 1 4 1 5 2 5
Proton
Number 31 32 33 34 35 36 37 38 39 40
Number of
Stable Isotopes 2 4 1 5 2 5 1 4 1 4
Proton
Number 41 42 43 44 45 46 47 48 49 50
Number of
Stable Isotopes 1 6 0 7 1 6 2 6 1 10
Proton
Number 51 52 53 54 55 56 57 58 59 60
Number of
Stable Isotopes 2 4 1 9 1 6 1 2 1 5
Proton
Number 61 62 63 64 65 66 67 68 69 70
Number of
Stable Isotopes 0 4 1 6 1 7 1 6 1 7
Proton
Number 71 72 73 74 75 76 77 78 79 80
Number of
Stable Isotopes 1 5 2 4 1 5 2 5 1 6
Proton
Number 81 82 83 84 85 86 87 88 89 90
Number of
Stable Isotopes 2 3
The odd-even alternation indicates a pairing of protons within the nucleus; perhaps the existence of alpha particle subsystems.
The average number of stable isotopes increases with proton number reaching a peak for proton number 50 (Tin) and declines generally thereafter, as shown in the table below.
Proton
Number
Range 0's 10's 20's 30's 40's 50's 60's 70's 80's 90's
Average
Number of
Stable Isotopes 1.7 2.3 2.8 3.0 3.4 3.7 3.2 3.3 1.1 0.0
The real test of the magic-ness of a number is whether it appears so in terms of the neutron number. The number of stable isotopes as a function of neutron number are:
Neutron
Number 1 2 3 4 5 6 7 8 9 10
Number of
Stable Isotopes 2 2 1 1 2 2 2 2 1 3
Neutron
Number 11 12 13 14 15 16 17 18 19 20
Number of
Stable Isotopes 1 3 1 3 1 3 1 3 0 5
Neutron
Number 21 22 23 24 25 26 27 28 29 30
Number of
Stable Isotopes 0 3 2 3 1 4 4 4 1 4
Neutron
Number 31 32 33 34 35 36 37 38 39 40
Number of
Stable Isotopes 1 3 1 3 0 2 3 7 1 4
Neutron
Number 41 42 43 44 45 46 47 48 49 50
Number of
Stable Isotopes 1 5 3 4 1 3 1 4 2 5
Neutron
Number 51 52 53 54 55 56 57 58 59 60
Number of
Stable Isotopes 2 4 3 4 2 3 2 3 1 3
Neutron
Number 61 62 63 64 65 66 67 68 69 70
Number of
Stable Isotopes 1 5 1 3 1 3 2 2 1 7
Neutron
Number 71 72 73 74 75 76 77 78 79 80
Number of
Stable Isotopes 4 5 1 5 2 4 2 4 3 3
Neutron
Number 81 82 83 84 85 86 87 88 89 90
Number of
Stable Isotopes 2 8 4 1 3 1 2 4 0 6
Neutron
Number 91 92 93 94 95 96 97 98 99 100
Number of
Stable Isotopes 2 4 1 4 1 6 1 5 2 4
Neutron
Number 101 102 103 104 105 106 107 108 109 110
Number of
Stable Isotopes 2 2 2 5 2 2 0 3 0 3
Neutron
Number 111 112 113 114 115 116 117 118 119 120
Number of
Stable Isotopes 2 2 1 2 1 2 3 1 3 3
Neutron
Number 121 122 123 124 125 126 127 128 129 130
Number of
Stable Isotopes 1 3 1 3 0 1 1 0 0 3
Neutron
Number 131 132 133 134 135 136 137 138 139 140
Number of
Stable Isotopes 0 0 0 0 0 3 1 1 1 0
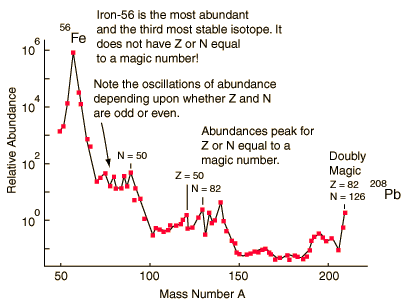
Note that Silicon is the element with atomic number 14 and it has three stable isotopes whereas the elements Aluminum and Phosphorus having atomic numbers 13 and 15, respectively, have only one stable isotope each. The isotope of silicon which is most prevalent has atomic weight 28=14+14. The binding energy per nucleon for silicon is however not exceptionally high, but it is the end result of a fusion process in stars called silicon burning, indicating a significant degree of stability.
-
Double magic
Nuclei which have neutron number and proton (atomic) numbers each equal to one of the magic numbers are called "double magic", and are especially stable against decay. Examples of double magic isotopes include helium-4, oxygen-16, calcium-40, calcium-48, nickel-48, nickel-78, and lead-208.
Double-magic effects may allow existence of stable isotopes which otherwise would not have been expected. An example is calcium-40, with 20 neutrons and 20 protons, which is the heaviest stable isotope made of the same number of protons and neutrons. Both calcium-48 and nickel-48 are double magic because calcium-48 has 20 protons and 28 neutrons while nickel-48 has 28 protons and 20 neutrons. Calcium-48 is very neutron-rich for such a light element, but like calcium-40, it is made stable by being double magic. Nickel-48, discovered in 1999, is the most proton-rich isotope known beyond helium-3. At the other extreme, nickel-78 is also doubly magical, with 28 protons and 50 neutrons, a ratio observed only in much heavier elements apart from tritium with one proton and two neutrons (Ni-78: 28/50 = 0.56; U-238: 92/146 = 0.63).
Magic number shell effects are seen in ordinary abundances of elements: helium-4 is among the most abundant (and stable) nuclei in the universe and lead-208 is the heaviest stable nuclide.
Magic effects can keep unstable nuclides from decaying as rapidly as would otherwise be expected. For example, the nuclides tin-100 and tin-132 are examples of doubly magic isotopes of tin that are unstable, and represent endpoints beyond which stability drops off rapidly.
In December 2006 hassium-270, with 108 protons and 162 neutrons, was discovered by an international team of scientists led by the Technical University of Munich having the half-life of 22 seconds. Hassium-270 evidently forms part of an island of stability, and may even be double magic.
-
Magic numbers are typically obtained by empirical studies; if the form of the nuclear potential is known then the Schrödinger equation can be solved for the motion of nucleons and energy levels determined. Nuclear shells are said to occur when the separation between energy levels is significantly greater than the local mean separation.
In the shell model for the nucleus, magic numbers are the numbers of nucleons at which a shell is filled. For instance the magic number 8 occurs when 1s1/2, 1p3/2, 1p1/2 energy levels are filled as there is a large energy gap between the 1p1/2 and the next highest 1d5/2 energy levels.
The atomic analog to nuclear magic numbers are those numbers of electrons leading to discontinuities in the ionization energy. These occur for the noble gases helium, neon, argon, krypton, xenon, radon and ununoctium. Hence, the "atomic magic numbers" are 2, 10, 18, 36, 54, 86 and 118.
In 2007, Jozsef Garai from Florida International University proposed a mathematical formula describing the periodicity of the nucleus in the periodic system based on the tetrahedron.
In 2010, an alternative explanation of magic numbers was given in terms of symmetry considerations. Based on the fractional extension of the standard rotation group, the ground state properties (including the magic numbers) for metallic clusters and nuclei were simultaneously determined analytically. A specific potential term is not necessary in this model.
-