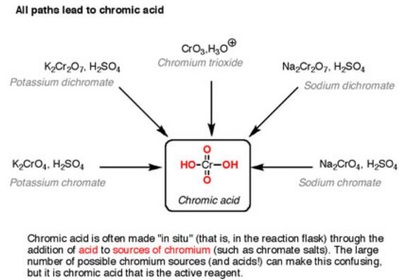
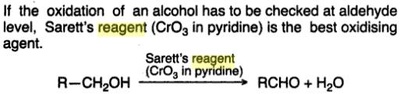
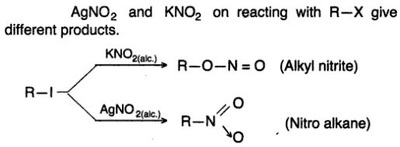
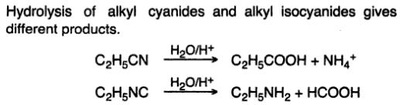
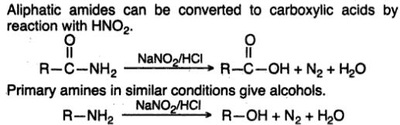
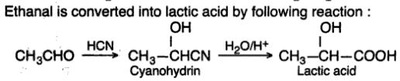
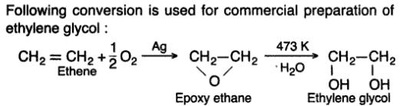
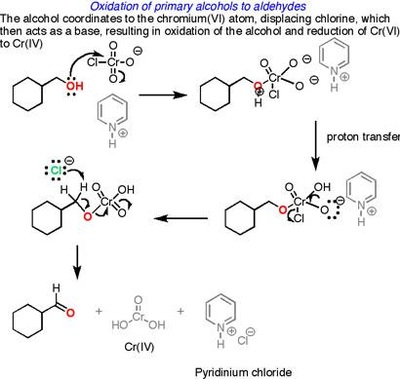
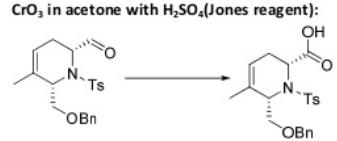
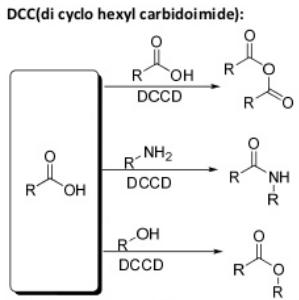
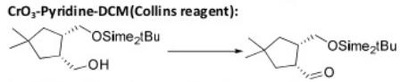
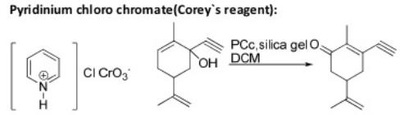
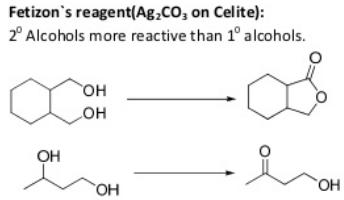
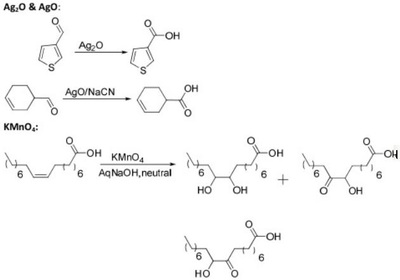
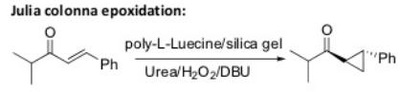
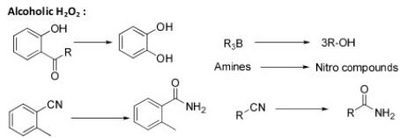
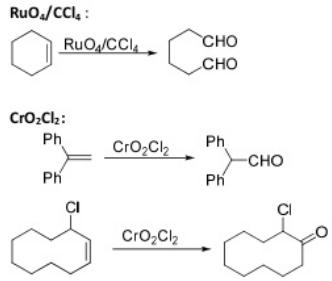
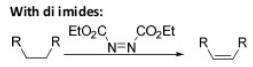
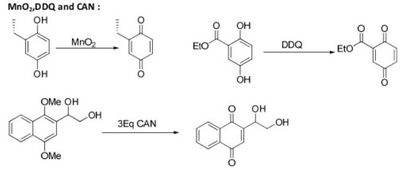
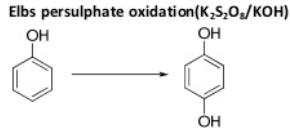
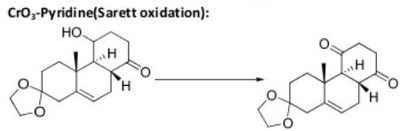
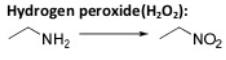
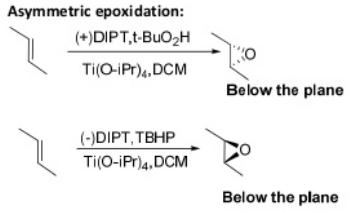
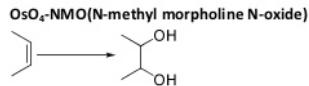
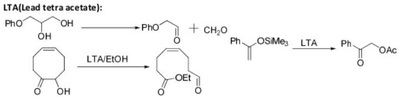
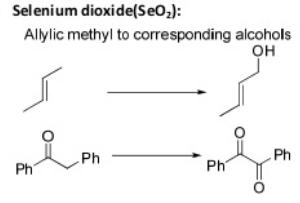
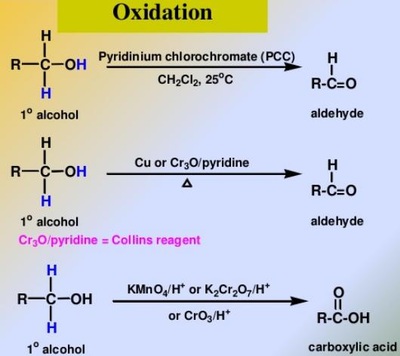
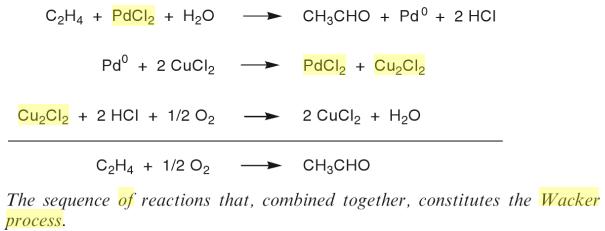
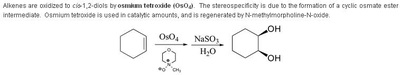
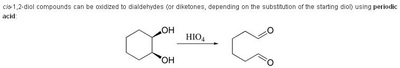
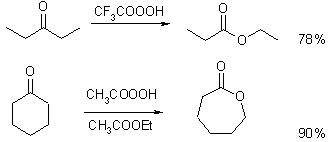
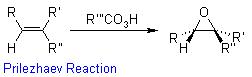
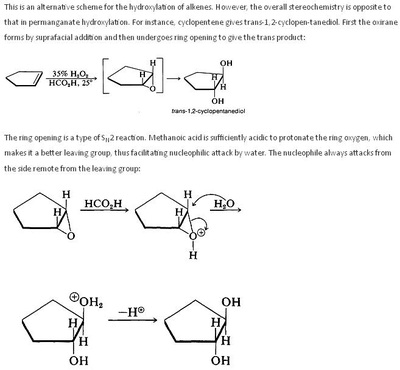
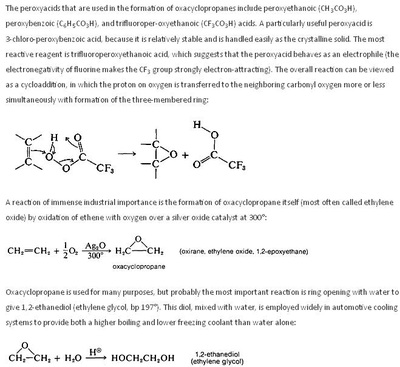
Oxidation Reactions in Organic Chemistry
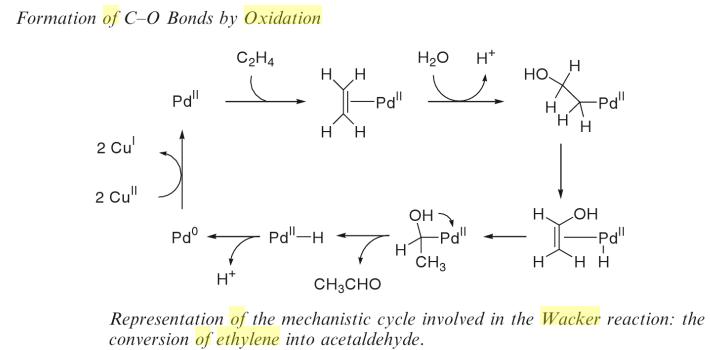
It had been known since 1894 that ehylene is oxidized to acetaldehyde by Palladium chloride.
The Wacker process or the Hoechst-Wacker process (named after the chemical companies of the same name) originally referred to the oxidation of ethylene to acetaldehyde by oxygen in water in the presence of a tetrachloropalladate(II) catalyst.
The development of the chemical process now known as the Wacker process began in 1956 at Wacker Chemie.
The same basic reaction is currently used to produce aldehydes and ketones from a number of alkenes with the Monsanto process for producing acetic acid.
This chemical reaction, a German invention, was the first organometallic and organopalladium reaction applied on an industrial scale. The Wacker process is similar to hydroformylation, which is also an industrial process and also leads to aldehyde compounds. The differences are that hydroformylation promotes chain extension, and uses a rhodium-based catalyst system. The Wacker process is an example of homogeneous catalysis. The palladium complex with ethylene is reminiscent of Zeise's salt, K[PtCl3(C2H4)] which is a heterogeneous catalyst.
At the time, many simple aliphatic compounds were produced from acetylene (as calcium carbide) but the construction of a new oil refinery in Cologne by Esso close to a Wacker site, combined with the realization that ethylene would be a cheaper raw-material prompted Wacker to investigate its potential uses.
As part of the ensuing research effort, a reaction of ethylene and oxygen over palladium on carbon in a quest for ethylene oxide unexpectedly gave evidence for the formation of acetaldehyde (simply based on smell). More research into this ethylene to acetaldehyde conversion resulted in 1957 in a gas-phase reaction patent using a heterogeneous catalyst.[3] In the meanwhile Hoechst AG joined the race and after a patent filing forced Wacker into a partnership
called Aldehyd GmbH. The heterogeneous process ultimately failed due to catalyst inactivation and was replaced by the water-based homogeneous system for which a pilot plant was operational in 1958. Problems with the aggressive catalyst solution were solved by adopting titanium (newly available for industrial use) as construction material for reactors and pumps. Production plants went into operation in 1960.
The Wacker process or the Hoechst-Wacker process (named after the chemical companies of the same name) originally referred to the oxidation of ethylene to acetaldehyde by oxygen in water in the presence of a tetrachloropalladate(II) catalyst.
The development of the chemical process now known as the Wacker process began in 1956 at Wacker Chemie.
The same basic reaction is currently used to produce aldehydes and ketones from a number of alkenes with the Monsanto process for producing acetic acid.
This chemical reaction, a German invention, was the first organometallic and organopalladium reaction applied on an industrial scale. The Wacker process is similar to hydroformylation, which is also an industrial process and also leads to aldehyde compounds. The differences are that hydroformylation promotes chain extension, and uses a rhodium-based catalyst system. The Wacker process is an example of homogeneous catalysis. The palladium complex with ethylene is reminiscent of Zeise's salt, K[PtCl3(C2H4)] which is a heterogeneous catalyst.
At the time, many simple aliphatic compounds were produced from acetylene (as calcium carbide) but the construction of a new oil refinery in Cologne by Esso close to a Wacker site, combined with the realization that ethylene would be a cheaper raw-material prompted Wacker to investigate its potential uses.
As part of the ensuing research effort, a reaction of ethylene and oxygen over palladium on carbon in a quest for ethylene oxide unexpectedly gave evidence for the formation of acetaldehyde (simply based on smell). More research into this ethylene to acetaldehyde conversion resulted in 1957 in a gas-phase reaction patent using a heterogeneous catalyst.[3] In the meanwhile Hoechst AG joined the race and after a patent filing forced Wacker into a partnership
called Aldehyd GmbH. The heterogeneous process ultimately failed due to catalyst inactivation and was replaced by the water-based homogeneous system for which a pilot plant was operational in 1958. Problems with the aggressive catalyst solution were solved by adopting titanium (newly available for industrial use) as construction material for reactors and pumps. Production plants went into operation in 1960.
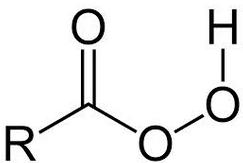
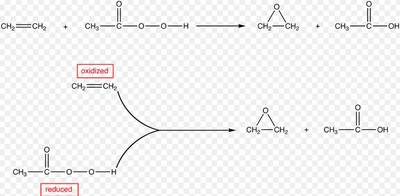
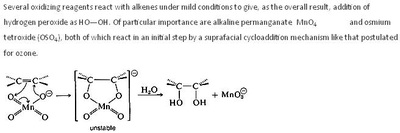
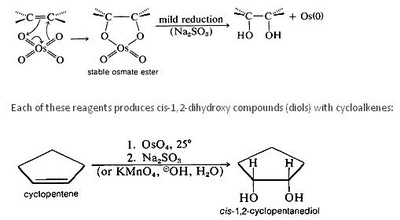
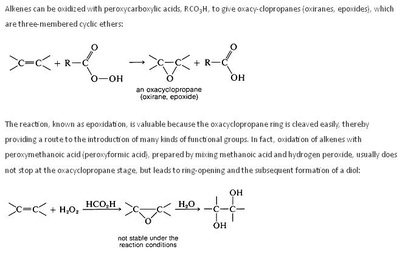
A peroxy acid (often spelled as one word, peroxyacid, and sometimes called peracid) is an acid which contains an acidic -OOH group. The two main classes are those derived from conventional mineral acids, especially sulfuric acid, and the organic derivatives of carboxylic acids. They are generally strong oxidizers.
Peroxymonosulfuric acid (Caro's acid) is probably the most important inorganic peracid, at least in terms of the scale. It is used for the bleaching of pulp and for the detoxification of cyanide in the mining industry. It is produced by treating sulfuric acid with hydrogen peroxide. Peroxyphosphoric acid (H3PO5) is prepared similarly.
Peroxymonosulfuric acid (Caro's acid) is probably the most important inorganic peracid, at least in terms of the scale. It is used for the bleaching of pulp and for the detoxification of cyanide in the mining industry. It is produced by treating sulfuric acid with hydrogen peroxide. Peroxyphosphoric acid (H3PO5) is prepared similarly.
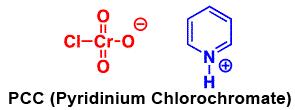
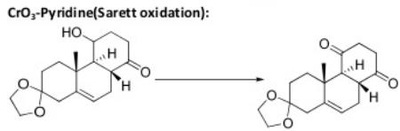
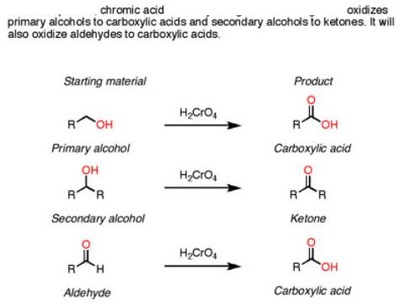
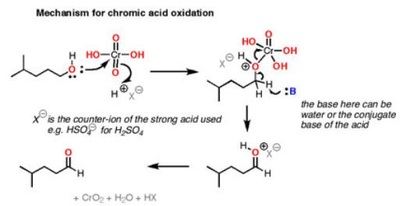
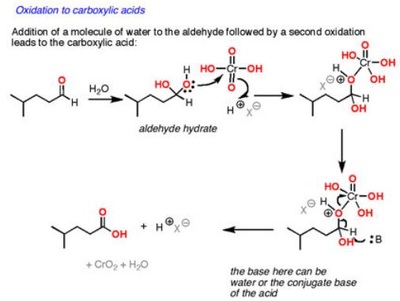
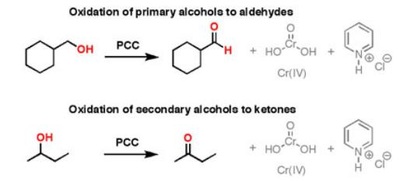
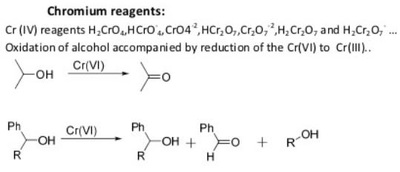
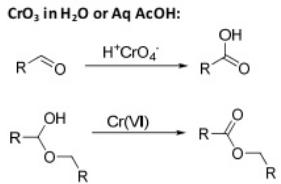
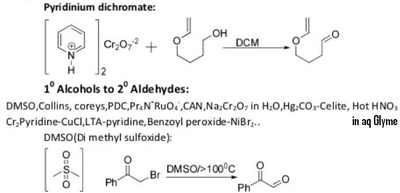
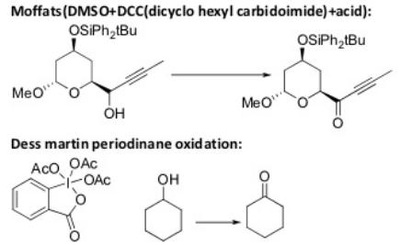
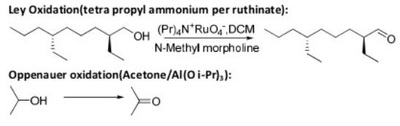
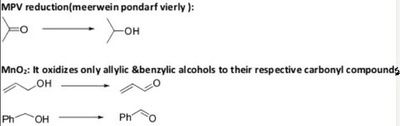
The pyridinium chlorochromate (PCC) and Swern oxidation reactions are useful for oxidizing primary alcohols to aldehydes. Further oxidation of the aldehyde to the carboxylic acid stage does not occur with these reagents, because the reactions are carried out in anhydrous (water-free) organic solvents such as dichloromethane, and therefore the hydrate form of the aldehyde is not able to form.
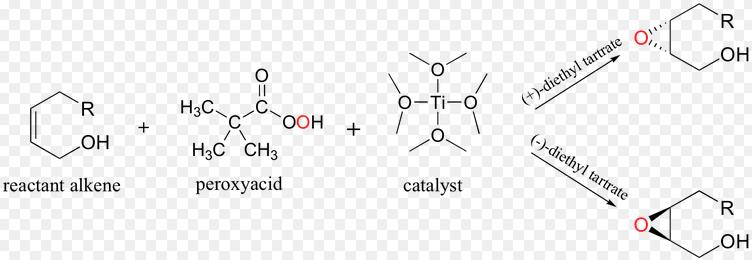
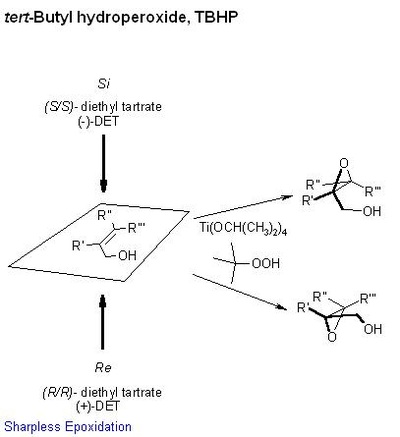
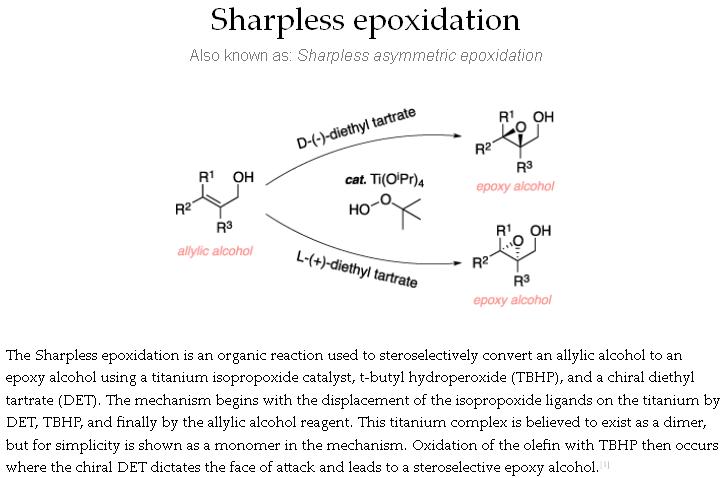
tert-Butyl hydroperoxide (tBuOOH) is an organic peroxide widely used in a variety of oxidation processes, for example Sharpless epoxidation. It is normally supplied as a 69-70% aqueous solution. Industrially, tert-butyl hydroperoxide is used as a starter of radical polymerization.
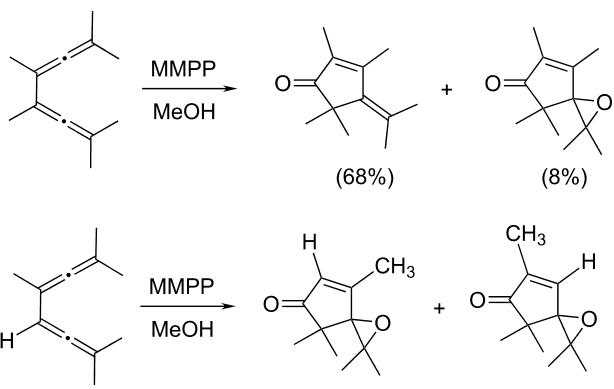
Magnesium monoperoxyphthalate (MMPP) is a water-soluble peroxy acid used as an oxidant in organic synthesis. Its main areas of use are the conversion of ketones to esters (Baeyer-Villiger oxidation), epoxidation of alkenes (Prilezhaev reaction), oxidation of sulfides to sulfoxides and sulfones, oxidation of amines to produce amine oxides, and in the oxidative cleavage of hydrazones. Due to its insolubility in nonpolar solvents MMPP has seen less use than the more widely used meta-chloroperoxybenzoic acid (mCPBA). Although work up procedures are more simply handled in polar solvents, usage of MMPP to oxidize nonpolar substrates in biphasic media combined with a phase transfer catalyst have been inefficient.[1] Despite this MMPP has certain advantages over mCPBA including a lower cost of production and increased stability.
MMPP is also used as the active ingredient in certain surface disinfectants such as Dismozon Pur. As a surface disinfectant MMPP exhibits a broad spectrum biocidal effect including inactivation of endospores. Its wide surface compatibility enables its use on sensitive materials, such as plastic and rubber equipment used in hospitals. Additionally MMPP has been investigated as a potential antibacterial agent for mouthwashes and toothpaste.
MMPP is also used as the active ingredient in certain surface disinfectants such as Dismozon Pur. As a surface disinfectant MMPP exhibits a broad spectrum biocidal effect including inactivation of endospores. Its wide surface compatibility enables its use on sensitive materials, such as plastic and rubber equipment used in hospitals. Additionally MMPP has been investigated as a potential antibacterial agent for mouthwashes and toothpaste.
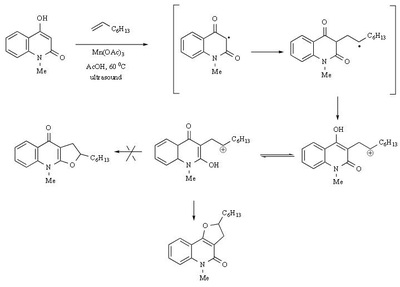
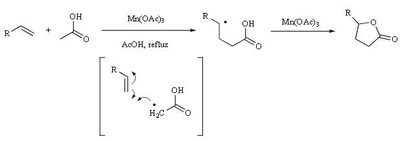
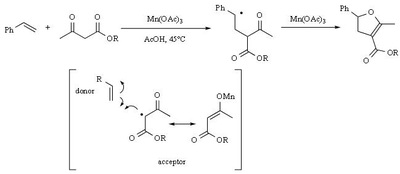
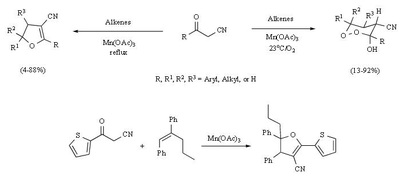
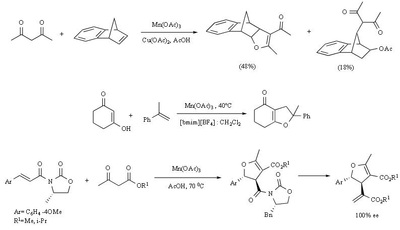
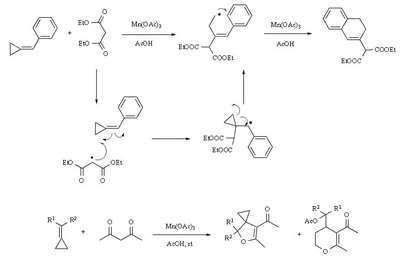
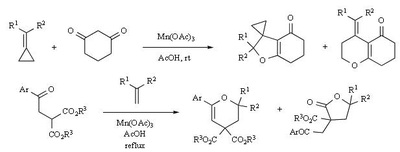
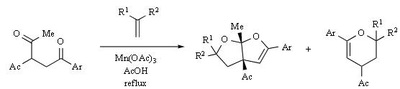
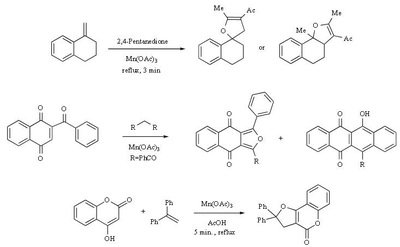
Mn(OAc)2 oxidation
Metal-promoted radical reactions have found widespread use in organic synthesis, in which one of the well-known examples of this application is the manganese(III) acetate mediated reactions. The exciting development in this area is beginning to show its true potential, as evidenced from the application of this methodology in strategy-level bond formation during the synthesis of complex molecules. Manganese(III) acetate dihydrate [ abbreviated herein as
Mn(OAc)3 ] mediated free radical reactions have emerged as important synthetic methods for a new bond formation and bond breaking. In view of its importance in organic synthesis, this present review highlights the application of Mn(OAc)3 promoted freeradical reactions in carbon-carbon, carbonheteroatom bond formation, and covers the literature since 1993.
Metal-promoted radical reactions have found widespread use in organic synthesis, in which one of the well-known examples of this application is the manganese(III) acetate mediated reactions. The exciting development in this area is beginning to show its true potential, as evidenced from the application of this methodology in strategy-level bond formation during the synthesis of complex molecules. Manganese(III) acetate dihydrate [ abbreviated herein as
Mn(OAc)3 ] mediated free radical reactions have emerged as important synthetic methods for a new bond formation and bond breaking. In view of its importance in organic synthesis, this present review highlights the application of Mn(OAc)3 promoted freeradical reactions in carbon-carbon, carbonheteroatom bond formation, and covers the literature since 1993.
volume strength of hydrogen peroxide
Volume strength is a unique measure of H2O2. It is the number of volumes of O2 liberated on decomposition of a unit volume of H2O2. ( at STP )
10 ml 5Vol H2O2, on decomposition liberates 10×5 = 50ml of O2.
Standard grade H2O2 sold to public is generally 10Vol H2O2.
For example: If 1cc of H2O2 liberates 100cc oxygen at S.T.P. The strength of H2O2 will be called 100 volumes H2O2.
-
"Volume strength" is a term unique to hydrogen peroxide. What it means is that if you have one volume strength H2O2 then one volume of it will generate, or liberate, one volume of oxygen. If the volume is one liter of H2O2, then one liter of oxygen will be liberated. If you have ten volume strength H2O2, then each volume, whether cc, liters, cubic feet, will liberate 10 times that volume of oxygen.
This strange term came to be used since H2O2 decomposes into water. Over time, it loses its oxygen and becomes weaker and weaker until it is finally just water. So even if it is say 10% H2O2 when fresh, a year later, it may just be plain water. So to tell what its strength was at any given time, you could take a certain volume of it, add a chemical to cause it to decompose, and measure the volume of oxygen given off to give its strength.
-
Question : The strength of H2O2 (in g/litre) in 11.2 volume solution of H2O2 is ?
2 moles of H2O2 = 22.4 L of oxygen at STP .
so, by that first of all u have to calculate the MOLARITY of the solution and use the formula
moles of solute / volume of solution
and the volume of solution is given as 11.2
and you need to calculate the no of moles.
and use the formula
mass/ molar mass , or given mass / molar mass and findout the no of moles,
THE STRENGTH CAN BE CALCULATED BY THE FOLLOWING FORMULA
11.2 * MOLARITY OF THE SOLUTION.
from the previous formula , you find molarity and apply it here , you will get the strength
-
Using a Reduction/Oxidation Reaction to Measure Mass Percent of Hydrogen Peroxide
Volume strength is a unique measure of H2O2. It is the number of volumes of O2 liberated on decomposition of a unit volume of H2O2. ( at STP )
10 ml 5Vol H2O2, on decomposition liberates 10×5 = 50ml of O2.
Standard grade H2O2 sold to public is generally 10Vol H2O2.
For example: If 1cc of H2O2 liberates 100cc oxygen at S.T.P. The strength of H2O2 will be called 100 volumes H2O2.
-
"Volume strength" is a term unique to hydrogen peroxide. What it means is that if you have one volume strength H2O2 then one volume of it will generate, or liberate, one volume of oxygen. If the volume is one liter of H2O2, then one liter of oxygen will be liberated. If you have ten volume strength H2O2, then each volume, whether cc, liters, cubic feet, will liberate 10 times that volume of oxygen.
This strange term came to be used since H2O2 decomposes into water. Over time, it loses its oxygen and becomes weaker and weaker until it is finally just water. So even if it is say 10% H2O2 when fresh, a year later, it may just be plain water. So to tell what its strength was at any given time, you could take a certain volume of it, add a chemical to cause it to decompose, and measure the volume of oxygen given off to give its strength.
-
Question : The strength of H2O2 (in g/litre) in 11.2 volume solution of H2O2 is ?
2 moles of H2O2 = 22.4 L of oxygen at STP .
so, by that first of all u have to calculate the MOLARITY of the solution and use the formula
moles of solute / volume of solution
and the volume of solution is given as 11.2
and you need to calculate the no of moles.
and use the formula
mass/ molar mass , or given mass / molar mass and findout the no of moles,
THE STRENGTH CAN BE CALCULATED BY THE FOLLOWING FORMULA
11.2 * MOLARITY OF THE SOLUTION.
from the previous formula , you find molarity and apply it here , you will get the strength
-
Using a Reduction/Oxidation Reaction to Measure Mass Percent of Hydrogen Peroxide
To learn more see the following links
http://zookeepersblog.wordpress.com/chemistry-organic-south-bangalore-skm-classes-for-iseet/
http://zookeepersblog.wordpress.com/reagents-of-chemistry/
http://zookeepersblog.wordpress.com/the-edges-and-pages-of-organic-chemistry-with-dj-daksha-and-janet/
http://zookeepersblog.wordpress.com/ncert-cbse-standard-12-haloalkanes-and-haloarenes-chapter-10-organic-chemistry/