Ruby LASER working
was asked in Karnataka COMEDK exam
was asked in Karnataka COMEDK exam
After Schawlow and Townes published their paper on the possibility of laser action in the infrared and visible spectrum it wasn't long before many researchers began seriously considering practical devices. Most experts were speculating that gases would be the first to lase in the optical and infrared.
It came as surprise that ruby was the first substance to produce laser action in the visible spectrum (Maiman, 1960).
The first working laser was built in 1960 by Maiman, using a ruby crytal and so called the Ruby laser. The ruby cylinder forms a Fabry-Perot cavity by optically polishing the ends to be parallel to within a third of a wavelength of light. Each end was coated with evaporated silver, one end was made less reflective to allow some radiation to escape as a beam.
Ruby belongs to the family of gems consisting of Al2O3 with various types of impurities. For example pink Ruby contains 0.05% Cr atoms.
Initially it was thought that broadband optical pumping would be inefficient but this was only for ions with very narrow resonances such as those in gases and plasmas. When ions are embedded in a solid, they can absorb radiation in a much wider band of wavelengths. Optical radiation of about 550 nanometers is absorbed by dilute population of Cr+3 ions lightly doped in a corundum crystal matrix (0.05% by weight Cr2O3 versus Al2O3 ) then a rapid non-thermal transition is made to a lower metastable level whose lifetime is 5 milliseconds. If the pump power exceeds a certain threshold, a population inversion can build up between the ground state and this metastable state. The laser's performance is greatly improved by enclosing it in an optical resonator.
Photo-pumped by a fast discharge flash-lamp, the first ruby lasers operated in pulsed mode for reasons of heat dissipation and the need for high pumping powers. Nelson and Boyle (1962) constructed a continuous lasing ruby by replacing the flash lamp with an arclamp.
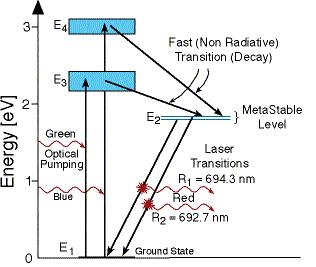
Ruby is an aluminum oxide (Al2O3) crystal, called sapphire, with a small amount of chromium oxide (Cr2O3) added to it. Sapphire is colorless and transparent, but the chromium doped crystal is pinkish-red in color because it strongly absorbs both in the green and in the blue. When this crystal is excited through the absorption of blue and/or green light it soon causes excitation of a meta-stable energy state of the chromium ion (Cr+++). After a typical lifetime of a few milliseconds this state de-excites to the ground state with the emission of a 694.3 nm photon, which is visibly red in color.
So, the primary role of the aluminum oxide crystal, aside from hosting the chromium ion, is to absorb the pump energy and to excite the ion through collisions.
The ruby lazer consists of a ruby rod . which is made of chromium doped ruby material. At the opposite ends of this rod there are two silver polished mirrors. Whose one is fully polished and other is partially polished. A spring is attached to the rod with fully polished end for adjustment of wave length of the lazer light. Around the ruby rod a flash light is kept for the pump input. The whole assembly is kept in the glass tube. Around the neck of the glass tube the Radio Frequency source and switching control is designed in order to switch on and off the flash light for desired intervals.
When we switch on the circuit the R.F operates. As a result the flash of light is obtained around the ruby rod. this flash causes the electrons within ruby rod to move from lower energy band towards higher energy band. The population inversion take place at high energy band and electrons starts back to travel towards the lower energy band. During this movement the electron emits the laser light. This emitted light travels between the two mirrors where cross reflection takes place of this light. The stimulated lazer light now escapes from partially polished mirror in shape of laser beam.
The spring attached with the fully polished mirror is used to adjust the wave length equal to λ/2 of lazer light for optimum laser beam. The switching control of the R.F source is used to switch on and off the flash light so that excessive heat should not be generated due to very high frequency of the movement of the electron.
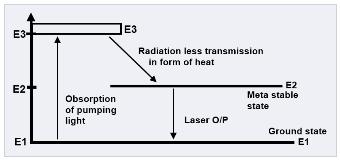
The above three level energy diagram show that in ruby lasers the absorption occurs in a rather broad range in the green part of the spectrum. This makes raise the electrons from ground state E1 to the band of level E3 higher than E1. At E3 these excited levels are highly unstable and so the electrons decays rapidly to the level of E2. This transition occurs with energy difference (E1 – E2) given up as heat (radiation less transmission). The level E2 is very important for stimulated emission process and is known as Meta stable state. Electrons in this level have an average life time of about 5m.s before they fall to ground state. After this the population inversion can be established between E2 and E1. The population inversion is obtained by optical pumping of the ruby rod with a flash lamp. A common type of the flash lamp is a glass tube wrapped around the ruby rod and filled with xenon gas. When the flash lamp intensity becomes large enough to create population inversion, then stimulated emission from the Meta stable level to the ground level occurs which result in the laser output. Once the population inversion begins, the Meta stable level is depopulated very quickly. Thus the laser output consists of an intense spike lasting from a few Nano sec to µsec. after stimulated emission spike, population inversion builds up again and a 2nd spike results. This process continues as long as the flash lamp intensity is enough to create the population inversion.
The Chromium ions absorb light at wavelengths around 545 [nm] (500-600 [nm]). As a result the ions are transferred to the excited energy level E3.
From this level the ions are going down to the metastable energy level E2 in a non-radiative transition. The energy released in this non-radiative transition is transferred to the crystal vibrations and changed into heat that must be removed away from the system.
A short while after the initial announcement of the first successful optical laser, other labs around the world jumped on the bandwagon trying out many different substrates and ions such as rare earths like Nd, Pr, Tm, Ho, Er, Yb, Gd even Uranium was successfully lased ! Many different substrates were tried such as Yttrium Aluminum Garnet (YAG), glass (which was easier to manufacture), CaF2. As manufacturing techniques improved these lasers rapidly made the transition from the lab bench to commercial applications.
Advantages of Ruby Lasers
From cost point of view, the ruby lasers are economical.
Beam diameter of the ruby laser is comparatively less than CO2 gas lasers.
Output power of Ruby laser is not as less as in He-Ne gas lasers.
Since the ruby is in solid form therefore there is no chance of wasting material of active medium.
Disadvantages of Ruby Laser
In ruby lasers no significant stimulated emission occurs, until at least half of the ground state electrons have been excited to the Meta stable state.
Efficiency of ruby laser is comparatively low.
Optical cavity of ruby laser is short as compared to other lasers, which may be considered a disadvantage.
Applications of ruby Laser
Due to low output power they are class-I lasers and so may used as toys for children’s.
It can be used in schools, colleges, universities for science programs.
It can be used as decoration piece & artistic display.
From this level the ions are going down to the metastable energy level E2 in a non-radiative transition. The energy released in this non-radiative transition is transferred to the crystal vibrations and changed into heat that must be removed away from the system.
A short while after the initial announcement of the first successful optical laser, other labs around the world jumped on the bandwagon trying out many different substrates and ions such as rare earths like Nd, Pr, Tm, Ho, Er, Yb, Gd even Uranium was successfully lased ! Many different substrates were tried such as Yttrium Aluminum Garnet (YAG), glass (which was easier to manufacture), CaF2. As manufacturing techniques improved these lasers rapidly made the transition from the lab bench to commercial applications.
Advantages of Ruby Lasers
From cost point of view, the ruby lasers are economical.
Beam diameter of the ruby laser is comparatively less than CO2 gas lasers.
Output power of Ruby laser is not as less as in He-Ne gas lasers.
Since the ruby is in solid form therefore there is no chance of wasting material of active medium.
Disadvantages of Ruby Laser
In ruby lasers no significant stimulated emission occurs, until at least half of the ground state electrons have been excited to the Meta stable state.
Efficiency of ruby laser is comparatively low.
Optical cavity of ruby laser is short as compared to other lasers, which may be considered a disadvantage.
Applications of ruby Laser
Due to low output power they are class-I lasers and so may used as toys for children’s.
It can be used in schools, colleges, universities for science programs.
It can be used as decoration piece & artistic display.