About Elements, their Naming, and Properties
Elements are divided into s-Block elements, p-Block elements, d-Block elements and f-Block elements
We know that electrons are arranged in the shells of atoms around the nucleus. These shells are arranged according to their energy and can be represented as K, L, M, and N.... The atomic shells can be further classified as s, p, d and f sub-shells. Each sub-shell is divided into orbitals which are denoted as s, p, d, and f-orbitals. The long form of the periodic table is also based in these orbital and sub-shells.
The s-block of the periodic table consists of the first two groups i.e the groups IA and IIA namely the Alkali Metals and the Alkaline Earth Metals along with Hydrogen and Helium.
s-block elements include hydrogen (H), helium (He), lithium (Li), beryllium (Be), sodium (Na), magnesium (Mg), potassium (K), calcium (Ca), rubidium (Rb), strontium (Sr), cesium (Cs), barium (Ba), francium (Fr) and radium (Ra). The Group IA of the Periodic Table consists of elements like Lithium (Li), Sodium (Na), Potassium(K), Rubidium (Rb), Cesium (Cs) and Francium (Fr) which are collectively known as Alkali Metals. These are so called because they form hydroxides on reaction with water which are strongly alkaline in nature.
All of the s-block elements are metals. In general, they are shiny, silvery, good conductors of heat and electricity and lose their valence electrons easily. In fact, they lose their trademark s orbital valence electrons so easily that the s-block elements are considered to be some of the most reactive elements on the periodic table. The Group IIA consists of elements like Beryllium(Be),Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba) and Radium(Ra) which are collectively known as Alkaline Earth Metals (except Beryllium) .They are so called because their oxides and hydroxides are alkaline in nature and these metal oxides are found in the earth's crust. In s-block elements the last electron enters the outermost s-orbital and as the s-orbital can accommodate only two electrons, that is why only two groups i.e group 1 & 2 belong to the s-block of the Periodic Table. Thus the outermost orbital of s block elements consists of one or two electrons and the orbital next to the outermost shell i.e the penultimate shell has either 2 or 8 electrons. This is the reason why the s-block elements show a fixed valency which depends on the number of electrons present in the outermost shell. The last element of both the groups i.e, Francium(Fr) and Radium (Ra) are radioactive and possess different properties than the other elements of the same group.
The periodic table can be divided into blocks; s, p, d and f according to their valence shell electronic configuration. The elements of s-block have their valence electron in s-sub shell. A first and second group of the periodic table is part of s-block. From 3rd group, the valence electrons are filled in d-sub shell therefore these elements are part of d-block and continue till 12th group of the periodic table. Form 13th group, again valence electrons are again filled in p-sub shell, therefore these elements are known as p-block elements. p-block elements are placed from 13th to 18th group of the periodic table. They include metals, non-metals and metalloids. This block is better explained group wise and each group is known with its 1st element such as carbon family, nitrogen family, boron family etc. Let’s have a look at some more features of p-block of elements.
The elements in column 1, known collectively as the alkali metals (except hydrogen), always lose their one valence electron to make a +1 ion. These metals are characterized by being silvery, very soft, not very dense and having low melting points. These metals react extremely vigorously with water and even oxygen to produce energy and flammable hydrogen gas. They are kept in mineral oil to reduce the chance of an unwanted reaction or worse, an unwanted explosion.
The elements in column 2, known as the alkaline earth metals (except helium), always lose their two valence electrons to make a +2 ion. Like the alkali metals, the alkaline earth metals are silvery, shiny and relatively soft. Some of the elements in this column also react vigorously with water and must be stored carefully.
S-block elements are common for being ingredients in fireworks. The ionic forms of potassium, strontium and barium make appearances in firework displays as the brilliant purples, reds and greens.
Francium is considered to be the most rare naturally occurring element on earth. It is estimated that there is only a few grams of Francium present on earth at a time. Francium has a very unstable nucleus and undergoes nuclear decay rapidly. As soon as it is created it vanishes.
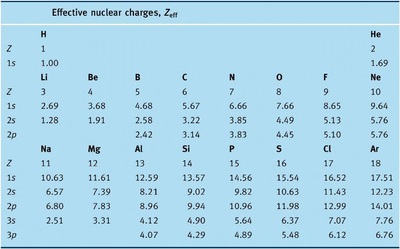
1) Electronic Configuration :- The general electronic configuration of s-block elements is ns 1 for Alkali metals and ns 2 for Alkaline earth metals where n = 2 to 7 . All the Alkali metals have one valence electron and these loosely held s-electron in the outermost valence shell of these elements makes them the most electropositive metals. The Alkaline Earth Metals have two electrons in the s-orbital of the valence shell. Like Alkali Metals, these elements are also electropositive ( Electronegativity value less than 1 ).
2) Metallic character :- All the Alkali metals are silvery white, soft and light metals. The Alkaline Earth metals, in general,are silvery white, lustrous and relatively soft but harder than the Alkali metals. Beryllium and magnesium appear to be somewhat greyish. The Metallic character increases as we go down both groups. Both the Alkali metals and the Alkaline Earth metals are highly malleable and ductile and have a very high tendency to loose electrons to form positive ions and hence they are highly electropositive ( Meaning in Pauling scale electronegativity values less than 1 ).
3) Atomic Density :- The Alkali metals and the Alkaline Earth metals both have low density. This is because they have large ionic size due to which their atomic nuclei are widely separated in their crystal lattices. The density increases down both groups.
4) Melting and Boiling Points :- The melting and boiling points of the Alkali metals are low indicating weak metallic bonding due to the presence of only a single valence electron in them. The melting and boiling points of Alkaline Earth metals are higher than the corresponding alkali metals due to their smaller sizes. The trend is, however, not systematic but it slightly decreases down the group.
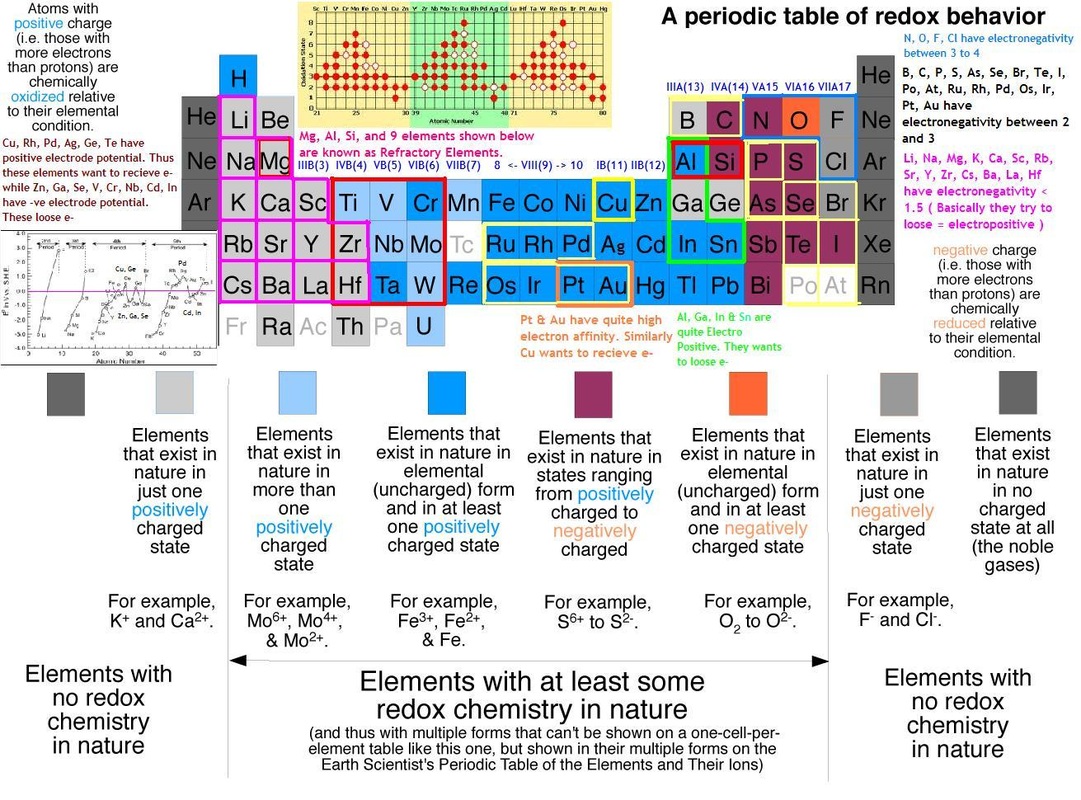
5) Oxidation state :- The Alkali metals show only +1 oxidation state, while Alkaline Earth metals show +2 oxidation state only.
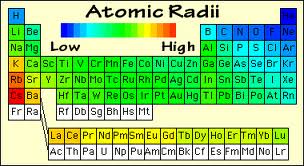
6) Atomic and Ionic radii :- Both the Alkali metals and the Alkaline Earth metals have large atomic and ionic radii. The Atomic and the Ionic radii increases as we move down both groups. But as we go from group I to group II in the same period the Atomic and the Ionic radii decreases.
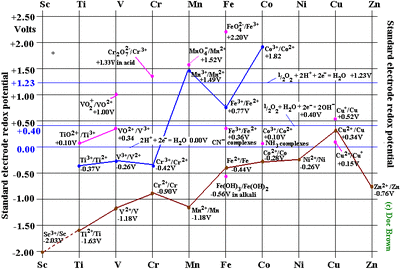
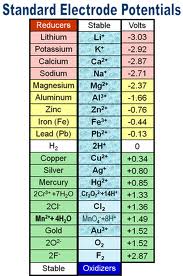
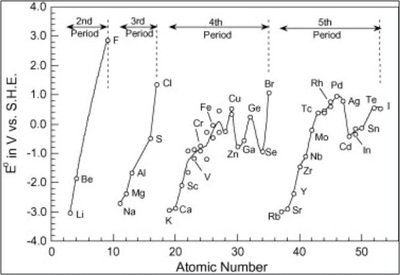
7) Electrode potential :- The Alkali metals are strong reducing agents. The Standard Electrode Potentials of all alkali metals lie between -2.7V and -3.0V, indicating a strong tendency to form cations in solution. The Alkaline Earth Metals also have negative values of their Standard Electrode potentials.
8) Ionisation Energies :- The ionization energies of the Alkali metals are considerably low and decreases down the group from Lithium (Li) to Cesium (Cs).This is due to the increasing size, increasing nuclear charge, and the outermost electron is very well screened from the nuclear charge. The Alkaline Earth metals have low ionization energies due to their large size of the atoms. Since the atomic size increases down the group, their ionization energies decreases down the group. But as we go from group I to group II in the same period Ionisation Energies increases. The first ionisation energies ( means the energy required to remove the first electron from the atom ) of the alkaline earth metals are higher than those of the corresponding Group I metals. This is due to their small size as compared to the corresponding alkali metals. But the second ionisation energies (means the energy required to remove the second electron from the atom) of the alkaline earth metals are smaller than those of thecorresponding alkali metals.
9) Magnetic Properties :- Alkali metals are attracted by the applied magnetic field and hence are Paramagnetic in nature whereas the Alkaline Earth metals are repelled by the magnetic field and hence are diamagnetic in nature.
10) Complex Formation :- Both the Alkali metals and the Alkaline earth metals show weak tendency to form complexes because they have no low energy vacant orbital available for bonding with lone pair of ligands.This is due to large size, low nuclear charge and poor ability to attract electrons.
11) Chemical Reactivity :- The alkali metals are highly reactive metals and the reactivity increases down the group. The reactivity is due to :-
(a) Low value of first ionization energy
(b) Large size
(c) Low heat of atomization
Reactivity of Alkali Metals :-
1) Alkali metals lose their luster very easily upon exposure to air.
2) Alkali metals react with water and form hydroxides along with liberation of hydrogen.
3) Alkali metals react with hydrogen and form ionic hydrides and these metal hydrides in turn react with water to give back the hydrogen.
4) Alkali metals react with halogens very easily forming halides.
5) All alkali metals are readily soluble in liquid ammonia.
6) The alkali metals are good reducing agents and the reducing property increases from Li to Cs.
7) Alkali metals can form alloys with other elements in the same period or with metals in other groups.
8) Alkali metals react with sulphur and phosphorus on heating to form sulphides and phosphides.
9) Alkali metals combine with mercury to form amalgams.
Reactivity of Alkaline Earth Metals :-
The alkaline earth metals are less reactive than the alkali metals.
Reactivity of alkaline earth metals increases as we move down the group.
1) Beryllium and magnesium are kinetically inert to oxygen and water because of the formation of an oxide film on their surface.
2) All the alkaline earth metals combine with halogen at elevated temperatures forming their halides.
3) All the elements except beryllium combine with hydrogen upon heating to form their hydrides.
4) The alkaline earth metals readily react with acids liberating dihydrogen.
5) Like alkali metals, the alkaline earth metals are strong reducing agents.
6) Like alkali metals, the alkaline earth metals dissolve in liquid ammonia to give deep blue black solutions forming ammoniated ions.
7) The alkaline earth metals burn in oxygen to form the monoxide.
8) The alkaline earth metals also form salts of oxoacids like carbonates,sulphates and nitrates.
9) They directly combine with carbon and form carbides .
12) Conductivity :- Both the Alkali metals and the Alkaline Earth metals are good conductors of heat and electricity.
13) Colour :- All the alkali elements are silvery white solid. When freshly cut, they have a bright lusture which quickly fades due to surface oxidation.The silvery luster of alkali metals is due to the presence of highly mobile electrons of the metallic lattice.
The alkaline earth metals, in general,are silvery white and lustrous .
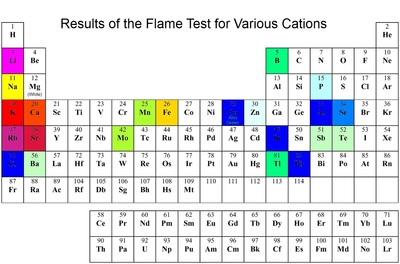
14) Flame coloration :- This property is due to the ease of excitation of the valence electrons. When elements or their compounds are introduced to flame, the electrons absorbs energy from the flame and gets excited to higher energy levels. When these electrons return to their ground state, they emit absorbed energy in form of visible light having characteristic wavelengths. Depending upon the wavelength of light emitted, different colours are imparted to the flame. Salts (generally chlorides) impart characteristic colours to the Bunsen flame.
The alkali metals and their salts impart a characteristic colour to flame.
For eg :-
Lithium - Crimson Red
Sodium - Golden Yellow
Potassium - Pale Violet
Rubidium - Violet
The alkaline earth metals also imparts colours to flame.
For eg :-
Strontium - Bright crimson
Calcium - Brick red
Barium - Apple green
Radium - Carmine – red
15) Photoelectric Effect :- Alkali metals (except Li) and the Alkaline Earth metals exhibit photoelectric effect (A phenomenon of emission of electrons from the surface of metal when light falls on them). The ability to exhibit photoelectric effect is due to low value of ionization energy of alkali and alkaline earth metals. Li does not emit photoelectrons due to high value of ionization energy.
Description of Helium (group I element) :-
Helium is named for the Greek god of the Sun, Helios. It was first detected as an unknown yellow spectral line signature in sunlight during a solar eclipse in 1868 by French astronomer Jules Janssen. Janssen is jointly credited with detecting the element along with Norman Lockyer. Jannsen observed during the solar eclipse of 1868 while Lockyer observed from Britain. Lockyer was the first to propose that the line was due to a new element, which he named. The formal discovery of the element was made in 1895 by two Swedish chemists, Per Teodor Cleve and Nils Abraham Langlet, who found helium emanating from the uranium ore cleveite. In 1903, large reserves of helium were found in natural gas fields in parts of the United States, which is by far the largest supplier of the gas today.
Helium is an element with symbol He and atomic number 2. It is a colorless, odourless, tasteless, non-toxic, inert and monatomic gas. It is the second lightest element and is the most abundant element in the universe. Helium is the least water soluble monoatomic gas. It's the less reactive element and doesn't essentially form chemical compounds. The density and viscosity of helium vapour are very low.It has low boiling point, low density and high thermal conductivity.Natural gases contain higher helium concentrations than the atmosphere.
Description of Hydrogen (group II element) :-
Hydrogen is an element with symbol H and atomic number 1. It is a colourless, odorless, tasteless, flammable and nontoxic gas atmospheric temperatures and pressures.It is the most abundant element in the universe. Hydrogen is the lightest of all gases.Hydrogen burns in air with a pale blue colour, almost invisible flame. Hydrogen has the highest combustion energy,This property makes it the fuel of choice for multi-stage rockets. Hydrogen has the lowest boiling point of any element except helium. Hydrogen was Discovered by H. Cavendish and named by A. Lavoisier .It's Density is about 0.08988 g/l . It's Melting point is about 14.01 K and Boiling point is about 20.28 K . It has dimagnetic magnetic properties.Hydrogen gas can form explosive mixtures with air. The destruction of the Hindenburg airship was an famous example of hydrogen combustion. Hydrogen is the only element that has different names for its isotopes i.e Deuterium , Protium and tritium . The electrolysis of water is a simple method of producing hydrogen. Hydrogen can be prepared in several different ways, but economically the most important processes involve removal of hydrogen from hydrocarbons.
SKMClasses Bangalore
____________________________________________________________________________
P Block elements
The p-block of the periodic table consists of the elements of groups 13,14, 15, 16, 17 and 18. These elements are characterised by the filling up of electrons in the outermost p-orbitals of their atoms. Some of these elements and their compounds play an important role in our daily life. For example:
Nitrogen is used in the manufacture of ammonia, nitric acid and fertilizers. Trinitrotoluene (TNT), nitroglycrine, etc., are compounds of nitrogen, which are used as explosives.
Oxygen present in air is essential for life and combustion processes.
Carbohydrates, proteins, vitamins, enzymes, etc., which contain chain of carbon atoms, are responsible for the growth and development of living organism.
P block chemistry is a little complicated compared to the s block chemistry. The reason behind this is that P block not only has non metals, but metalloids and metals too.
Every group, the boron group, Carbon group, Nitrogen group, Chalcogens, halogens and the noble gases have different physical and chemical properties.
Some of the common properties of 'P' block elements are
1. Atomic size
Atomic size of all elements in the 'P' block, decreases as we move from IIIA to VIIA.When we move down a group, say, for example, from carbon to lead in group IV A, the elements increase in size, due to the additional shell added.
2. Ionization energy
Ionization energy, or the energy to remove an electron from the outermost shell of an element, increases as we move along from IIIA to VIIA. It is maximum for a noble gas because noble gases have completely filled configuration. Ionization energy decreases as we move down a group. Some elements at the bottom of a group like Lead, tin, Thallium, Bismuth, etc. behave almost as like metals with very low ionization energies.
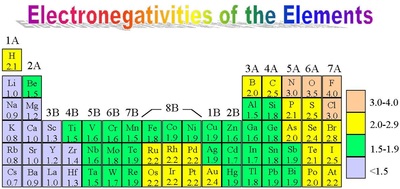
3. Electronegativity
The property of acquiring an electron or the ability to withdraw electrons from an electropositve element increases as we move from III A to VII A.
Elements of VII A have maximum electronegativity, with fluorine being the most electronegative atom present, due to its smaller size.
4. Allotropy
Allotropy is a phenomenon by which one element can exist in many forms. Most elements of IV A, V A, and VI A show allotropy.
For example, phosphorus exist in many forms like red phosphorus, white phosphorus, etc. Similarly, many forms of sulfur like monoclininc sulfur, rhombic sulfur, etc., are known. There are many forms of carbon too, like graphite, diamond, etc. Halogens do not show allotropy.
5. Catenation
Catenation is the ability to form compounds in which the atoms are linked to each other in chains or rings.
Carbon has the greater tendency to combine with other carbon atoms to form quite large carbon structures.
Other elements like silicon undergo catenation in IV A group. V A group also exhibits this property. Nitrogen and phosphorus have a tendency to form M
-M links.
Catenation can be seen in the case of Oxygen group too. S8 is an example of catenation.
Catenation is not shown among halogens and noble gases.
6. Chemical Properties of P block elements
Since P block elements are all non metals, covalent bond is seen in compounds such as hydrides, oxides, halides, etc.
The usual trends (vertical as well as horizontal) in various properties observed in the s-block are observed in this block, too. As we move from top to bottom through a vertical column (group) some similarities are observed in the properties. However, this vertical similarity is less marked in the p-block than that observed in the s-block, especially in groups 13 and 15; vertical similarity is increasingly shown by the later groups. As far as
the horizontal trend is concerned, the properties vary in a regular fashion as we move from left to right across a row (period).
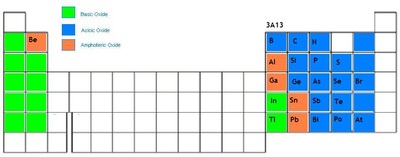
Elements belonging to the group 13 ( i.e group IIIA ) to group 17 (i.e group VIIA ) of the periodic table along with the group 18 i.e the zero group elements together form the p-block of the periodic table.
In the p-block elements the last electron enters the outermost p orbital. They have 3 to 8 electrons in the outermost shell. As we know that the number of p orbitals is three and, therefore, the maximum number of electrons that can be accommodated in a set of p orbitals is six. Hence there are six groups of p-block elements in the periodic table numbering from 13 to 18. The First group of the p-block i.e the group IIIA is commonly called as Boron group,the second group i.e the group IVA is called Carbon group,the third group i.e the group VA is called Nitrogen group, the fourth group i.e the group VIA is called Chalcogens ,the fifth group i.e the group VIIA is called Halogens and the sixth group i.e the zero group or group 18 is called Inert or Noble gases group. In the p-block all the three types of elements are present, i.e. the Metals,Non-Metals and Metalloids .The zigzag line in the p-block separates all the elements that are metals from those that
are non-metals. Metals are found on the left of the line, and non-metals are those on the right. Along the line we find the metalloids. Due to the presence of all types of elements the p-block show a lot of variation in properties.
Classification of elements in the p-block of the Periodic Table :-
The elements in the p-block of the periodic table consists of all types of elements i.e metals , non-metals and metalloids.
a) Metals :-
1)Aluminium, 2)Gallium, 3)Indium, 4)Thallium, 5)Tin, 6)Lead, 7)Bismuth .
b)Non-Metals :-
1) Helium, 2) Carbon, 3) Nitrogen, 4) Oxygen, 5) Fluorine, 6) Neon, 7) Phosphorus, 8) Sulphur, 9) Chlorine, 10) Argon, 11) Selenium, 12) Bromine, 13) Krypton, 14) Iodine, 15) Xenon, 16) Radon.
c) Metalloids :-
1)Boron, 2) Silicon, 3) Germanium, 4) Arsenic, 5) Antimony, 6) Tellurium, 7) Polonium, 8) Astatine.
Characteristic Properties of elements in p-block of Modern Periodic Table :-
1) Electronic Configuration :- The general valence shell electronic configuration of p-block elements is ns 2 np 1-6 (except for He). The inner core of the electronic configuration may, however, differ.
The General electronic configuration shown by elements from group13 to 18 of p-block is as given below :-
Group 13 (Boron family) :- ns 2 np 1 .
Group 14 (Carbon family) :- ns 2 np 2 .
Group 15 (Nitrogen family) :- ns 2 np 3 .
Group 16 (Oxygen family) :- ns 2 np 4 .
Group 17 (Halogen family) :- ns 2 np 5 .
Group 18 (Noble gases) :- ns 2 np 6 (except Helium) .
The general electronic configuration of Helium is 1s 2. Due to their distinct electronic configuration p-block elements show a lot of variation in properties.
2) Metallic Character :- As stated earlier p-block contains all types of elements i.e metals,non-metals and metalloids . The p-block is the only region of the periodic table to contain metalloids .The non metallic character decreases down the group whereas there is a gradual increase in non-metallic character from left to right in the p-block. The metallic character tends to increase down each group whereas it decreases as we go from left to right across a period. In fact, the heaviest element in each p-block group is the most metallic in nature.
3) Atomic Density :- The Atomic Density of elements in p-block increases down the group, this is due to increase in the size of the atom down the group.
Whereas it decreases as we move from left to right across the period,this is due to the decrease in atomic size of all elements in the p- block across the period. Of all the elements, aluminum is of very low density and is widely used as a structural material.
4) Melting and Boiling points :- The Melting and Boiling points gradually increases down the group because the molecular mass increases down the group and hence the intermolecular forces increases.
5) Oxidation state :- The p-block elements show a variable oxidation state . The oxidation states increases as we move from left to right in the periodic table. The maximum oxidation state shown by a p-block element is equal to the total number of valence electrons . According to this, the oxidation states
shown by different groups is as follows :-
Boron family (Group 13) :- +3
Carbon family ( Group 14) :- +4
Nitrogen family (Group 15) :- +5
Oxygen family ( Group 16) :- +6
Halogen family (Group 17) :- +7
Noble gases ( Group 18) :- +8
But in addition to this p-block elements may also show other oxidation states which normally , but not necessarily , differ from total number of valence electrons by unit of two. The other oxidation state two unit less than the group oxidation state shown by different groups is as follows :-
Boron family (Group 13) :- +1
Carbon family ( Group 14) :- +2 , -4
Nitrogen family (Group 15) :- +3, -3
Oxygen family ( Group 16) :- +4, +2, -2
Halogen family (Group 17) :- +5, +3, +1, -1
Noble gases ( Group 18) :- +6, +4, +2 .
However, the relative stabilities of these two oxidation states i.e the group oxidation state and the other oxidation state two unit less than the group oxidation state , may vary from group to group .
6) Atomic and Ionic radii :- As we move down the group in the p-block one extra shell than the preceding element gets added into the next element. This ultimately increases the atomic and the ionic radius of every next element down the group , which finally shows that the atomic and the ionic radii increases down the group.The trend is not same across the period. As we move from left to right in a period the Atomic radii and the Ionic radii of p-block elements decreases . The Atomic radius increases greatly from Boron to Aluminum.This increase is due to greater screening effect caused by the eight electrons present in the penultimate shell.
7) Electrode Potential :- The p-block elements generally have a positive electrode potential. It generally decreases down the groups.
For eg. Consider the electrode potentials of the halogen group :-
Fluorine = 2.87 V
Chlorine = 1.36 V
Bromine = 1.09 V
Iodine = 0.53 V
From the above analytical data we can say that the electrode potential
in the p-block decreases down the groups .
8) Ionisation Energies :- The p-block elements have high ionization potentials. The ionisation energies of p-block elements increases from left to right in a period due to increasing effective nuclear charge.
According to the general trends the ionisation energy values decreases down the group but do not decrease smoothly as expected. Non-metals have high Ionisation Energies than metals. It is maximum for a noble gas because noble gases have completely filled configuration. Some elements at the bottom of a group like Lead, Tin, Thallium, Bismuth, etc. behaves almost as a metal with very low ionization energies.
9) Magnetic Properties :- The elements Radon, Astatine, Iodine and Polonium of the p-block are Non-Magnetic in nature. The element Tin is Paramagnetic and the rest all elements of the p-block are Diamagnetic in nature.
10) Complex Formation :- The smaller size and the greater charge of the elements of different groups of p-block enable them to have a greater tendency to form complexes than the s-block elements. The complex formation tendency decreases down the group as the size of the atoms increases down the group.
11) Chemical Reactivity :- The Chemical Reactivity of elements in the p-block increases as we move from left to right in a period. But as we move down in a group the chemical reactivity of elements decreases down the group.
i) Reactivity of Noble gases :-
All the orbitals of the noble gases are completely filled by electrons and it is very difficult to break their stability by the addition or removal of electrons. Thus the noble gases exhibit very low chemical reactivity. Because of their low reactivity noble gases, are often used when an nonreactive atmosphere is needed, such as in welding.
Preceding the noble gas family there are two chemically important groups of non-metals. They are the halogens (Group 17) and the chalcogens (Group 16).
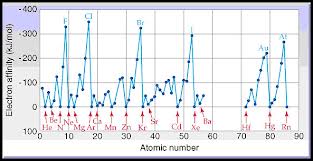
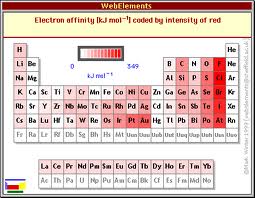
These two groups of elements have high electron gain enthalpies and can readily add one or two electrons forming an anion to attain the stable noble gas configuration thus showing good chemical reactivity.
ii) Reactivity of Halogens :-
a) All halogens are naturally found in a combined state.
b) Fluorine reacts readily with almost any substance coming in contact with it.
c) Chlorine, Bromine, and Iodine are progressively less reactive but still form compounds with most other elements, especially metals.
d) All the halogens are strong oxidising agents.The halogens oxidize other Substances, but themselves get reduced.
e) All halogens combine directly with sodium to form sodium halides.
f) All halogens react with red phosphorus to form phosphorus halides.
g) Halogens react readily with alkali metals forming salts.
h) The presence of Chlorine,Bromine and Iodine can be tested by the treatment of acidified silver nitrate solution.
iii) Reactivity of group VIA Elements ( Chalogens ) :-
a) As we approach to the right-hand side of the periodic table, similarities among the elements within a group become greater. This is true for the group VIA. Except Polonium, which is radioactive and usually omitted from all discussions.
b) All members of the group VIA form X 2– ions when combined with highly electropositive metals.
c) The tendency to be reduced to the - 2 oxidation state decreases significantly from top to bottom.
d) At ordinary temperatures and pressures, oxygen is a gas. It exists in either of the two allotropic forms :- O 2 , which makes up 21 percent of the earth's atmosphere, or O 3 (ozone), which slowly decomposes to O 2 .
e) The ozone itself absorbs longer-wavelength ultraviolet radiations , preventing these harmful rays from reaching the earth's surface which would otherwise increase the probability of human skin cancer and can also cause other environmental problems.
f) The Compounds of selenium and tellurium are of little commercial importance as they are toxic.
iv) Reactivity of Metalloids :-
a) The chemical reactivity of the metalloids depends on the substance with which it is reacting. For example:- Boron behaves as a nonmetal when reacting with sodium, but it acts as a metal when reacting with fluorine .
b) Thus from the above example we can say that Metalloids show variable chemical properties.
c) They act like non-metals when they react with metals whereas they act like metals when they react with non-metals.
d) Due to their low electronegativity, they are usually oxidized in reactions. The oxides of metalloids are usually amphoteric .
v) Reactivity of group VA Elements :-
a) All the group VA elements form trihydrides when reacted with hydrogen.
b) The reactivity decreases down the group.
c) The elements in the group VA either form trioxides or pentoxides when reacted with oxygen.
d) Also they form trihalides or pentahalides when reacted with halogens.
e) All the group VA elements react with metals to form binary compounds.
f) The most important compounds of the group VA elements are those of nitrogen and phosphorus.
g) Nitrogen and phosphorus are most commonly used as fertilizer.
vi) Reactivity of group IIIA Elements :-
1) Unlike groups IA and IIA, none of the group IIIA elements react directly with hydrogen to form hydrides.
Also all the group IIIA elements react with halogens to form trihalides instead of simply halides like group IA and IIA elements.
vii) Reactivity of group IVA elements :-
a) Carbon have a ability to form strong bonds with other carbon atoms and thus form a tremendous variety of organic compounds
b) In the +4 oxidation state lead acts as a strong oxidizing agent, gaining two electrons and after gaining electrons it gets reduced to the +2 oxidation state.
c) Also in the +4 oxidation state lead forms covalent compounds and bonds strongly to carbon.
d) Besides the metals themselves, some tin and lead compounds are of commercial importance. For eg :- Tin(II) fluoride (stannous fluoride), is added to some toothpastes to inhibit dental cares.
e) Lead is also found in two main commercial applications. One, the lead-acid storage batteries used to start cars and the other is in the automobile fuel.
12) Conductivity :- The conductivity of elements in p-block increases down the group. Generally the metals in the p-block are good conductors of heat and electricity whereas the non-metals are poor conductors of heat and electricity. The conductivity of metalloids lies in between the metals and non-metals.
13) Colour :-
Colour of group IIIA elements :-
All the group IIIA elements are silvery solids except boron which is brown solid.
Colour of group IVA elements :-
Carbon is black in colour whereas silicon and germanium have reddish brown or dull grey or black colour. Lead has a bluish-white colour.
Colour of group VA elements :-
Nitrogen is a colourless.
Phosphorus exists in white colour.
Arsenic is found in yellow and grey solid form.
Antimony is found in a amorphous grey form.
Bismuth is silvery white in colour .
Colour of group 16 elements :-
Oxygen is a gas and is colourless .
Sulphur is pale yellow in colour.
Tellurium is Silvery-white in colour .
All the Halogens are coloured. They have following colours :-
Fluorine :- Pale yellow.
Chlorine :- Greenish yellow.
Bromine :- Reddish brown.
Iodine :- Violet black.
Noble Gases have following colours :-
Helium is red, Neon is orange, Krypton is purple whereas Xenon is white in colour. Radon is colourless.
Helium is used for filling air ships and weather balloons because they are light and non-inflammable.
Neon is used as Beacon lights.
Argon is used for filling electric bulbs because of its inert nature.
Krypton is used in discharge tubes, filament lamps and decorative sign tubes.
Radon finds its use in the treatment of cancer along with radium. It is used in radiotherapy.
14) Flame colouration :- Not all but a few p-block elements impart characteristic colour to the flame. Arsenic impart Blue colour to the flame. Boron impart Bright green Colour to the flame. Copper (I) impart Blue colour whereas Copper(II) (non-halide ) impart Green colour to flame whereas Copper(II) ( halide ) impart Blue- green colour to flame. Indium and selenium impart Blue colour to flame. Phosphorus impart Pale bluish green colour whereas Lead impart Blue/White colour to flame. Antimony and Tellurium impart Pale green colour. Thallium impart Pure green colour to flame.
SKMClasses Bangalore
____________________________________________________________________________
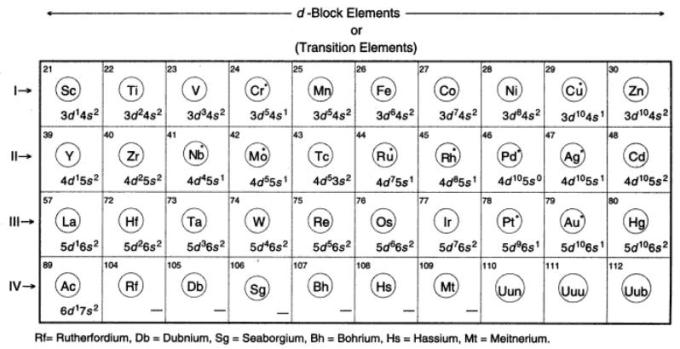
d Block Elements
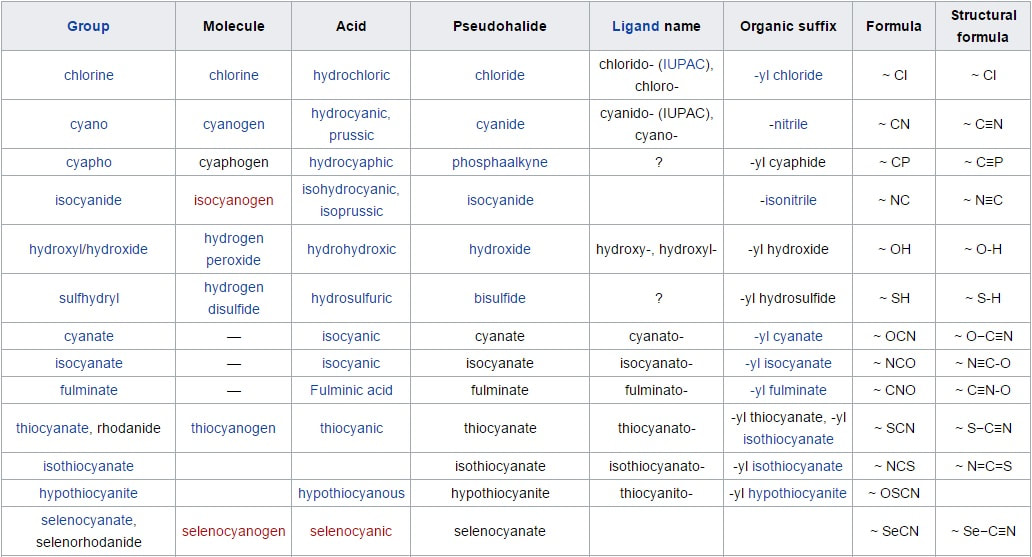
The elements in the groups from 3 to 12 or The elements in the groups IB, IIB, IIIB, IVB, VB, VIB, VIIB and, VIIIB of the periodic table are called as d-block elements. The 4th, 5th, 6th and 7th period consists of 10 elements each and are called as d-block elements. Thus, In all there are 40 d-block elements. The d-block elements have partially filled d-orbitals. They are placed in between the s-block and p-block elements in the periodic table. They are also called as Transition elements. The name " Transition " originates from their place between s and p-block elements in the periodic table. The d-block elements are all metals with their last two shells incompletely filled. The elements which have partially filled d-orbitals either ground state or in one or more of their ions, are called d-block elements or outer transition elements. Their properties are intermediate between s-block elements and
p-block elements. They are more electropositive than p-block elements but less electropositive than s-block elements. They form ionic compounds in their lower oxidation state and covalent compounds in higher oxidation states. They are all metals and have electronic configuration ns2, (n-1)d1 to 10 Characteristic Properties of elements in d-block of Modern Periodic Table :-
The properties of d-block elements are intermediate between s-block and p-block elements.
1) Electronic Configuration :- The general electronic configuration of d-block elements is ns 2 ( n - 1 ) d1 to 10 . But in the elements of the 12th group i.e Zn, Cd and Hg ,the incoming electron occupy the ns shell instead of ( n - 1 ) d subshell which are already saturated and they should therefore, be excluded from d-block elements , But, since they posses properties similar to those of d-block elements, for all practical purposes they are placed with d-block elements.
The d-block elements are classified into 4 series:-
a) 3d series containing 10 elements :-
They have incomplete 3d-orbital. Elements starting from Sc (21) to Zn (30) are present in these series.
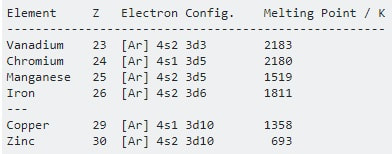
For first series (Sc z=21) to (Zn z=30)
Sc (z=21): 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d1.
Ti (z=22) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d2.
V (z=23) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d3.
Cr (z=24) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s1 , 3d5.
Mn (z=25) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2 ,3d5.
Fe (z=26) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2 ,3d6.
Co (z=27) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2 ,3d7.
Ni (z=28) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2 ,3d8.
Cu (z=29) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s1 ,3d10.
Zn (z=30) : 1s2, 2s2, 2p6, 3s2, 3p6, 4s2 ,3d10.
b) 4d series containing 10 elements :-
They have incomplete 4d-orbital. Elements starting from Y (39) to Cd(48) are present in these series.
c) 5d series containing 10 elements :-
They have incomplete 5d-orbital. Elements starting from La (57) to Hf (72) to Hg (80) are present in these series.
d) 6d series containing 10 elements :-
They have incomplete 6d-orbital. This is the incomplete series.
2) Metallic Character :- All the d-block elements have 1 or 2 electrons in their outermost shell and thus all are metals. All are hard,ductile and malleable solids with strong bonding due to their greater effective nuclear charge and large number of valence electrons ( except Hg which is liquid ) .
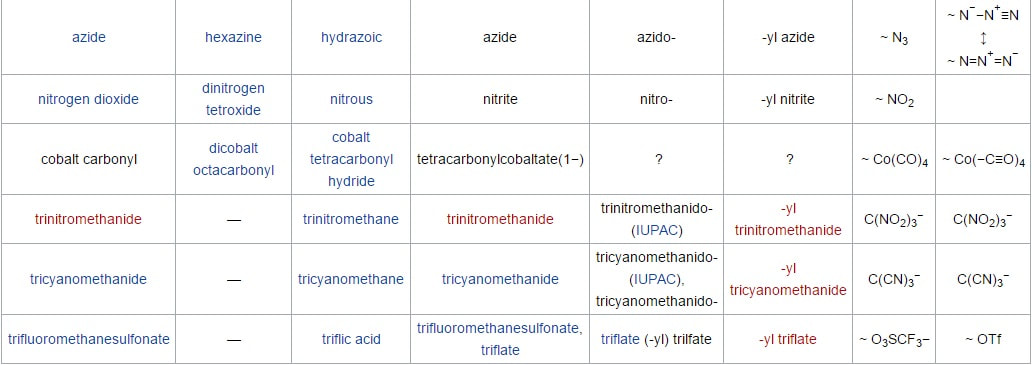
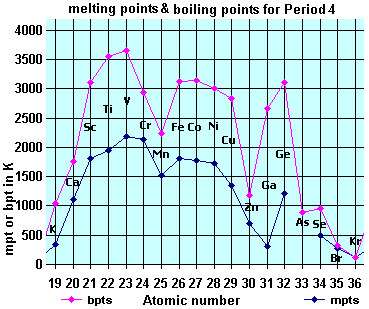
3) Atomic and Ionic Radii :- The Atomic and Ionic Radii of transition elements are smaller than their corresponding s-block elements and are greater than their corresponding p-block elements. The Atomic and Ionic Radii of transition elements for a given series show a decreasing trend for first 5 elements and then becomes almost constant for next 5 elements of the series. For eg :- In 3d series atomic radius decreases from Sc(21) to Mn(25) and then becomes constant for next five, i.e. Fe(26) to Zn(30). This is due to the combined effect of increasing effective nuclear charge and increasing screening effect along the period.
4) Atomic volume and density :- The size decreases along the period and therefore atomic volume also decreases along the period. Atomic volumes are smaller than group 1 and 2 members, i.e. s-block elements. The density,however,increases along the period.
5) Boiling and Melting points :- All the transition elements have high M.P. and B.P. , as compared to s-block elements due to their strong metallic bonding and unpaired d-electrons. M.P. of transition elements of a given series increases on moving from left to right in a period and attains a maximum value and after that M.P. goes on decreasing towards the end of period. These higher values are due to small atomic radii of transition elements which provides greater inter atomic forces of attraction. However Zn, Cd, Hg have relatively low values of M.P. and B.P. Due to their completely filled d-orbitals.
Melting and boiling point of these elements are very high (except Zn). These higher values are due to small atomic radii of transition elements which provides greater inter atomic forces of attraction. They are very hard .
6) Ionisation Energy :- The Ionisation energy of d-block elements lies in between the of s-block and p-block elements. Thus they are more electropositive than p-block elements and less electropositive than s-block elements. Since smaller being atomic size, thus high Ionisation Energy is noticed for transition elements. Ionization Energy values first increases upto Mn(25) and then becomes irregular or constant due to irregular trend of atomic size after Mn(25) in 3d series,also similar trend is noticed in 5d and 6d series. However Zn, Cd, Hg have abnormally high values of Ionization Energy on account of greater stability of s-subshell.
7) Oxidation state :- The Transition elements show variable oxidation state in their compounds. Reason for this variable oxidation state is that there is a very small energy difference in between (n-1)d and ns orbitals. As a result , electrons of (n-1)d orbitals as well as ns-orbitals take part in bond formation. Variation in oxidation state is related to their electronic configuration.
For e.g :-
Ti : +2, +3, +4.
Cr : +1, +2, +3, +4, +5, +6.
Mn : +2, +3, +4, +6, +7.
Fe : +2, +3.
Cu : +1, +2
8) Standard Electrode Potential and Reducing properties :- Expect Cu and Hg, the standard reduction potential of d-block elements in acid solution is generally negative, but however Cu and Hg are having positive electrode potential. The d-block elements are not good reducing agents in comparision to s-block elements due to their high heats of sublimation , high ionisation potential and low heats of hydration of their ions.
9) General Chemical Reactivity :- The d-block elements have been unreactive in comparision to s-block elements due to their high ionisation energy, high heats of sublimation and low heats of hydration.
But however Transition metals vary widely in their chemical reactivity. Many of them are sufficiently electropositive to dissolve in mineral acids, although a few are 'noble' ,that is, they areunaffected by simple acids. The metals of the first series with the exception of copper are relatively more reactive and can be oxidised.
Transition metals form alloys among themselves. The alloys of transition metals are hard and have high melting point as compared to other metal.
10) Conductivity :- All the Transition elements are good conductors of heat and electricity.
11) Colour :- Except zinc all the transition metals complex ions are colourless due to presence of unpaired electrons.
The colours exhibited by some transition metal ions are :-
1. Cu2+ (Blue).
2. Fe2+ (Green).
3. Ti3+ (Purple).
4. Mn3+ (Violet).
5. Fe3+ (Yellow).
12) Flame colouration :-The d-block elements impart no characteristic colour to the flame. We know that when an electron gains energy, it gets excited to a higher energy level, and when it looses that energy, it again comes back to the same level, by giving out some amount of radiation. This released energy appears in the form of colour. The d-block elements do not impart colour to the flame as their electron don't jump to the higher energy level when brought in the flame.
SKMClasses Bangalore
____________________________________________________________________________
f Block Elements
14 elements from atomic number 58 (Cerium) to 71(Lutetium) are called as Lanthanides and the another 14 elements from atomic number 90 (Thorium) to 103 ( Lawrencium ) are called as Actinides. These 28 elements are arranged in the 2 series placed below the Modern Periodic Table and the elements in these 2 series are collectively called as f-block elements. These elements have outermost three shells incomplete . In these elements the last or differentiating electron enters in to the f-orbital of anti-penultimate shell or the 3rd shell from the outermost shell,Thus the f-orbital is partially filled and hence they are called as f-block elements. They are also called as " Inner Transition elements ". They are so called because the last electron enters in the f-orbital of anti-penultimate shell, which is inner and these elements are present between strongly electropositive s-block elements and strongly electronegative p-block elements . The general electronic configuration of the f-block elements is given as :-
(n-2) f1-14, (n-1) s2 , p6 , d0-1, ns2 .
F- block elements are also called Inner transition elements. The last electron of these elements enters the 'f' sub shell. They are mostly radioactive elements. They are present beneath the main periodic table, in the form of two rows, one row belonging to lanthanides and the other row belonging to actinides.
Lanthanides
The group of 14 elements from Cerium(58) to Lutetium(71) are placed alongwith lanthanum in IIIB group and in the 6th period of the periodic table in a series below the periodic table called as Lanthanides.They are also called as Lanthanoids or Lanthanones and are represented as Ln . In Lanthanoids the last or differentiating electron enters into 4f orbital of the anti-penultimate shell and hence the series of 14 elements in lanthanoids is called as 4f series.
Europium and gadolinium has half-filled f-orbital. Ytterbium and lutetium has fully filled f-orbital.
Properties of Lanthanides :-
1) Electronic Configuration :- The General electronic configuration of Lanthanides is [Xe] 4f1–14 5d1 6s2 .
The complete electronic configuration of Lanthanides can be given as 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6 . The Lanthanoids have partially filled 4f-orbital.But, However ,the first member Lanthanum and the last member Lutetium of the lanthanide series have no partially filled 4f-orbital and they should therefore, be excluded from Lanthanoids, But, since they posses properties similar to those of Lanthanoids, for all practical purposes they are placed with Lanthanoids . We know that 4f orbital is filled only after completing 5s, 5p and 6s orbitals. It is found that when the f-orbital contains 0, 7 or 14 electrons it is stable. The electronic configuration of Lanthanum which is followed by 14 lanthanides is 2,8,18,18,9,2 . In Lanthanum 4f subshell is vacant and 5d subshell contains one electron. Thus in Lanthanides 4f orbitals are successively filled,means from Cerium(58) to Lutetium(71) ,the Additional electron should occupy vacant 5f orbital and 5d orbital should remain singly filled up. Thus the electronic configuration of Lanthanides should be 2,8,18,19-32,9,2 .
But in observed electronic configuration , 5d1 electron gets shifted to 4f orbital and 5d orbital remains vacant. It is because energies of 5d and 4f orbitals are closely similar.
2) Metallic Character :- Lanthanides are silvery white metals having tensile strength.
3) Atomic Density :- Lanthanides have lighter density values ranging from 6.77 to 9.74 gm/cm3 . Density increases with increase in atomic number.
4) Melting and Boiling Points :- These elements posses high M.P. and B.P. , but no regularity is seen with the increase in atomic number.
5) Oxidation state :- All Lanthanides show a common stable oxidation state +3 . It is because there are two electrons in the outermost shell and one electron in next to outermost shell i.e penultimate shell. Some Lanthanides show +2 and +4 oxidation states,these are observed in those elements , which by losing electrons attain the stable 4f0 ,4f7 and 4f14 configuration. Only +3 state is exhibited by La, Gd and Lu which is due to extract-ability of empty, completely half filled or completely filled 4f orbitals. Along with +3, other two oxidation states, +2 and +4 are exhibited by some members of this series.
Example: Eu and Yb can acquire +2 state as it provides them with extra stability.
The Ce4+ ion is stale and it can convert easily to Ce3+ thereby making the 4+ ion a very strong oxidizing agent.
6) Atomic and Ionic radii :- In Lanthanide series ,there is regular decrease in atomic size as well as atomic radii as the atomic number increases from Cerium to Lutetium. This decrease in size of atoms and ions is known as Lanthanide Contraction. On moving from Cerium to Lutetium , the atomic radii decreases from 165 pm to 156 pm and the ionic radii decreases from 103 pm to 85 pm.
7) Electrode Potential :- Due to Lanthanide Contraction the value of standard electrode potential increases regularly from La to Lu.
8) Ionisation Energies :- Lanthanides have very low ionisation energies. Ionisation energy values are similar to alkaline earth metals , particularly calcium. Due to low ionisation energies , Lanthanides are highly electropositive in nature.
9) Magnetic Properties :- Elements with paired electrons does not show any magnetism due to cancellation of the opposite spins due to pairing. Lanthanide ions having unpaired electrons and hence are paramagnetic ,while those having all the orbitals with paired electrons are diamagnetic in nature . Lanthanum and Lutetium have all paired electrons and hence are diamagnetic in nature.
10) Complex Formation :- Lanthanides do not have much tendency to form complexes due to their low charge density because of their large size. However, they form complexes with few chelating agents such as EDTA. The tendency to form complexes increases with increase in atomic number.
11) Chemical Reactivity :- As all the Lanthanides show a similar electronic configuration and common +3 oxidation state, they show similar Chemical Reactivity. They react with carbon, hydrogen, oxygen and sulfur and form carbides, hydrides, oxides and sulphides respectively. The oxides formed are M2O3 oxides. They react with water and form insoluble hydroxides. The oxides and hydroxides react with CO2 and form carbonates, M2(CO3)3.
a) All lanthanides react readily upon exposure to air and tarnish.
b) They readily dissolve in hot water liberating hydrogen. They can also dissolve in cold water.
c) They react with nitrogen and hydrogen forming the corresponding nitrides and hydrides.
d) Lanthanides react with other non-metals such as halogens, sulphur, phosphorus, carbon and silicon and form corresponding compounds.
e) The high oxidation potentials indicate their strong electro positive nature to act as strong reducing agents.
f) Lanthanides react with acids and liberate hydrogen.
12) Conductivity :- Lanthanides are good conductors of heat and electricity.
13) Colour :- All Lanthanides are silvery white metals . The trivalent lanthanide ions are coloured both in solid state and in aqueous solution,this colour change is seen only in case of cations. The colour of a cation depends on the number of unpaired f electrons.
Lanthanides with either half-filled or completely filled orbitals are colourless.
In moving along the lanthanide series from Ce(58) to Lu (71) a regular decrease in the size of the atom/ion with increase in atomic number is observed.
This decrease in size is called the Lanthanide contraction. In this series the size of Lanthanum is maximum and that of Lu, lutetium is minimum. The decrease is size, though continuous, is not regular.
Reason for Lanthanide Contraction
The cause of lanthanide contraction can be traced to the imperfect shielding of one 4f electron by another in the same sub-shell. On moving along the lanthanide series the number of 4f electrons increases by one unit at each step and the imperfect shielding increases, causing the contraction in electron cloud of the 4f sub-shell. Ionic radii changes from 1.06 Α to 0.85A.
Important Consequences
The lanthanide contraction plays a significant role in the chemistry of lanthanides. The important consequences of it are:
There is a steady decrease in ionic size.
There is a slight increase in electronegativity of the trivalent ions.
The Eo values for M3+ + 3e → M(g) increases regularly from Lanthanum -252V to 2.25 V for Lutetium.
Since there is a very small change in the size of ions in the lanthanide series and there is no change in the outermost shell, there is a close
resemblance in chemical properties.
Lanthanide contraction plays a significant role in the chemistry of lanthanides and heavier transition elements.
The atomic radii of 5d transition elements are very close to those of the corresponding 4d transition elements.
Due to this the crystal structure and other properties of lanthanides are very similar.
Applications of Lanthanides
Metallurgical applications: Some of the alloys of lanthanide elements find important metallurgical applications as reducing agents. Example: Misch meals (Ce- 30 to 35%)
Ceramic applications: Ce(III) and Ce(IV) oxides find use in glass polishing powders whereas Nd and Pr oxides are extensively used in coloring glass and
in the production of standard light filters.
Catalytic applications: Some lanthanide compounds are used as catalysts. Example: Cerium phosphate is used in petroleum cracking as a catalyst.
Electronic applications: The ferromagnetic garnets of 3Ln2O3.5Fe2O3 type are used in microwave devices.
Nuclear applications: These elements and some of their compounds are used in nuclear control devices, shielding devices and fluxing devices. Sm - 140,
Eu - 153, Gd- 155, Gd- 157 and Dy - 164 are some of the important isotopes used in nuclear technology.
Actinides
The group of 14 elements from Thorium(90) to Lawrencium(103) are placed alongwith Actinum in IIIB group and in the 7th period of the periodic table in a series below the periodic table called as Actinides.They are also called as Actinoids or Actinones and are represented as An . In Actinoids the last or differentiating electron enters into 5f orbital ,hence the series of 14 elements in Actinoids is called as 5f series. All Actinides are radioactive in nature.
Elements beyond Uranium are prepared synthetically in labouratory through nuclear reactions. The starting element for this purpose is always Uranium. Hence the elements from Neptunium to Lawrencium are called Transuranic elements ar Urenides.
Properties of Actinides :-
1) Electronic Configuration :-The general electronic configuration of Actinides is [Rn] 5f1-14 6d0-1 7s2 . The Actinoids have partially filled 5f-orbital.
The electronic configuration of Actinum which is followed by 14 Actinides is 2,8,18,32,18,9,2 . In Actinum 5f subshell is vacant and 6d subshell contains one electron. Thus in Actinides 5f orbitals are successively filled,means the Additional electron should occupy vacant 5f orbital and 6d orbital should remain singly filled up. Thus the electronic configuration of Actinides should be 2,8,18,32,19-32,9,2 .
But since it is not sure whether the last electron enters the 5f orbital or the 6d orbital as both have the same energy. Hence, there are two views regarding the electronic configuration of actinides.
According to Seaborg view, the 5f orbital is filled from thorium, while in Dawson view the 5f orbital is filled from uranium and are called uranides.
2) Metallic Character :- Actinides are silvery metals .
3) Atomic Density :- All Actinides except Thorium and Americium have high Atomic density.
4) Melting and Boiling Points :- These elements posses high M.P. and B.P. than the Lanthanides , but no regularity is seen with the increase in atomic number.
5) Oxidation state :- All Lanthanides show a common stable oxidation state +3 . The most stable oxidation state for first 3 elements
Thorium,Protactinium and Uranium is +4 , +5 and +6 .
Plutonium(Pu)shows all oxidation states from +3 to +7 but most stable is +4.
Americium shows oxidation state from +2 to +6.
6) Atomic and Ionic radii :- In the Actinide series ,there is regular decrease in atomic size as well as atomic radii as the atomic number increases from Actinium to Lawrencium. The size of ions decreases gradually along the series because extra charge of the nucleus is poorly shielded by f-electrons. It is called as Actinide Contraction and is similar to Lanthanide Contraction.
7) Electrode Potential :- Due to Actinide Contraction the value of standard electrode potential increases regularly from Ac to Lw.
8) Ionisation Energies :- Actinides have lower ionisation energies than lanthanides because 5f is more effectively shielded from nuclear charge than 4f.
Due to low ionisation energies , Actinides are highly electropositive in nature.
9) Magnetic Properties :- All Actinides have unpaired electrons and hence are paramagnetic in nature.
10) Complex Formation :- Actinides have higher tendency to form complexes than Lanthanides. It is because of their higher charge and smaller sizes of cations . Halides of actinides form complexes with alkali metals . Theses elements form chelates with organic compounds such as EDTA and oxime.
11) Chemical Reactivity :- Actinides are highly reactive metals in fine state and very strong reducing agents.
A few properties are given below.
a) They react with boiling water to give oxide and hydride.
b) They combine with most of non-metals at moderate temperature.
c) All these metals are attacked by HCl acid but the effect of nitric acid is very small.
d) They react with tarnish in air forming oxide coating.
12) Conductivity :- Actinides are good conductors of heat and electricity.
13) Colour :- Actinide ions are coloured . It depends upon the number of electrons in 5f orbitals. Ions having no electron in f orbital or 7 electrons in 5f orbital are colourless. The ions with 2 or 6 electrons in 5f orbitals are coloured in both the crystalline and in solution state due to f-f transition of electrons.
We know that the periodic table is divided into four blocks, depending upon the sub shell the last electron enters. The arrangement of elements in the periodic property is such that some properties follow a definite pattern.
The repetition of the elements with similar properties in the order of increasing atomic number as in the periodic table is called as Periodicity of properties.
Cause of periodicity
The properties of an element are reflected in its electronic configuration. In the long form of the periodic table, atoms having similar outermost electronic configuration appear after regular intervals. Therefore, the properties are repeated after a regular interval. All elements belonging to a particular group have similar electronic configuration and hence they exhibit similar properties.
Atomic size: Size of an atom Decreases along the period
Along a Group Increases with addition of a main orbital
SKMClasses Bangalore
_____________________________________________________________________________________________________________________
Various Details of Chemical Elements, origin of their names and uses are given here
Tc – Technetium – ( p – 43, M – 98 ) ( Radioactive ) The first element in the periodic table which is radioactive. So lightest Radioactive and Naturally Radioactive elemement. Medically used for identifying blockages in the body.
( Left of Ru – Ruthenium ). Tc was synthesized.
!
Uses of Tantalum
Ta – Tantalum – ( p – 73, M – 181 ) Ta was named after “ Torments of Tantalus “ which mean useless futile work. Tantalum was discovered after lot of try and lot of hard work ( Many false predictions in between ). Is used for making capacitors that are used in Mobile phones.
!
Nb – Niobium – ( p – 41, M – 93 ) Nb was discovered along with Tantalum. Niobium was daughter of Tantalus so the name! Niobium as an element and compounds have very similar properties as Vanadium and Tantalum.
!
F – Fluorine – ( p – 9, M – 19 ) F was last of the nonmetals discovered / separated in free state. Named after Greek Fluros meaning very destructive. Almost 30 scientists lost their lives and many more sustained burns and loss of laboratory equipments while trying to purify Fluorine. O2F2 , HF are some of the compounds.
!
Li – Lithium – ( p – 3, M – 7 ) From Greek Lithios meaning stone. Li was discovered in mineral kingdom while Na ( Sodium ) and K ( Potassium ) in plant kingdom. All Alkali elements are Silvery white Solid. When freshly cut has a bright luster. Li is more harder and lighter than other Alkali metals. Not affected by air easily and does not loose its lusture even after melting. Li reacts with H2O slowly to liberate H2. Li does not react with O2 below 0 deg C. Li when burnt in air gives monoxide Li2O while other alkali metals gives peroxides or super-oxides. Li2O is much less basic than oxides of other alkali metals. LiOH is much weaker base than NaOH or KOH. Li is the only Alkali metal which directly reacts with N2 to give Li3N. LiOH decomposes on heating to give Li2O. Hydroxides of other alkaline metals do not decompose. LiHCO3 is known in solution but not in solid state. While bicarbonates of other Alkali metals are known in solid state. Li2CO3 is less stable compared to carbonates of other Alkali metals. On heating gives li2O + CO2. LiNO3 on heating gives mixture of NO2 and O2, 4LiNO3 -> 2Li2O + 4NO2 + O2 while nitrates of other alkali metals yield only O2, 2NaNO3 -> 2NaNO2 + O2 Li2SO4 is the only alkali metal sulphate which does not form double salts e.g. alum. Li reacts with Br very slowly while all other alkali metals react violently. LiF, Li3PO4, Lithium Oxalate Li2C2O4, Li2CO3 are sparingly soluble in water. The corresponding salts of other alkali metals are freely soluble in water. LiCl is highly deliquescent ( becoming liquid by absorbing moisture from the air ) and soluble in Alcohol and pyridine. LiCl separates out from aqueous solutions as hydrated crystals LiCl.2H2O whereas Chlorides of other alkali metals are insoluble in organic solvents and do not form hydrated crystals under ordinary temperatures. Li when heated in NH3 forms imide Li2NH while other alkali metals form amides MNH2 Li shows lot of similarity with Mg known as diagonal relationship due to similarity in size, electronegativity and high polarizing power ( Ionic Potential ). ( Ionic Chrage / ( Ionic Radius )^2 ) [ Cations with large Ionic potentials have a tendency to polarize the anions and to give partial covalent character to compounds ] Li gives Crimson Red colour in flame test. Highest reducing power of Li in solution is due to its large heat of hydration.
!
Si – Silicon – ( p – 14, M – 28 ) The most abundant element on the Earth’s crust ( after oxygen ). Si is 28% of Earth’s crust. Latin name Silicium comes from Silex meaning hard stone. CO2 and SiO2 are acidic while GeO2, SnO2 and PbO2 are amphoteric in nature.
!
Al – Aluminium – ( p – 13, M – 27 ) 3rd most abundant element in Earth’s crust. Diaspore Al2O3.H2O Gibbsite Al2O3.3H2O Bauxite Al2O3.2H2O Cryolite Na3AlF6 Alunite K2SO4.Al2(SO4)3.4Al(OH)3 Torquoise AlPO4.Al(OH)3.H2O is usually blue coloured due to presence of copper phosphate. Felspar KalSi3O8 . It is a good reflective material so used in front coating of mirrors in an experiment in contrast to normal bathroom mirrors where the reflective coating is in the back. Aluminium foils are widely used for packing medicines, or food etc. In 19 th century when it was isolated for the first time, it was very costly and was displayed in museums.
!
Th – Thorium – ( p – 90, M – 232 ) Named after Thor. The ancient Scandinavian God of Thunder. Thorium in air burns very intensely. When mixed with glass gives very high Refractive index and very low dispersion, so used for making high quality lenses. Solid Solutions of Thorium Di-oxide and Yttrium oxide is transparent like glass for visible region. With a melting point around 2200 C this is used for peep windows for high temperature furnaces.
!
V – Vanadium – ( p – 23, M – 51 ) Named after Godess Vanadis. Scandinavian Godess of Beauty. It is used to make certain types of Catalysts. There are certain mushrooms which are rich in Vanadium.
!
Xe – Xenon – ( p – 54, M – 131 ) From Greek Xenos meaning stranger.
!
Hf – Hafnium and Re – Rhenium were last to be discovered even though these are stable. Hafnium oxide is being used as top layer of some computer chips and shows better properties than some other oxides which are being used. Hf ( p – 72, M – 178.5 ) and Re ( p – 75, M – 186.2 ) Rhenium is used in the alloy form in steel blades of aircrafts, as it gives good high temperature and stress properties. Rhenium is used as a catalyst. The name Rhenium is derived from river Rhine. Rhenium is one of the most rarest of the elements. Similar to Tungsten its boiling point is close to 6000 C. The only element which can form 8 different oxides. Rhenium is the only element that can form negatively charged univalent ions.
!
Sg – Seaborgium – ( p – 106, M – 263 ) The first element named after a living person, Glenn Seaborg who headed the team to make 6 artificial elements.
!
O – Oxygen – ( p – 8, M – 16 ) Oxus means acid. Gennas means maker. So Oxygen was “acid maker”. Oxygen differs from other elements of group 6A due to small size, High electronegativity, and non availability of d-orbitals in the valence shell. Oxygen is a gas while other elements are solid. O2 is diatomic while S8, Se8 etc are complex molecules. Oxygen is 2 nd most electronegative element and thus highly non-metallic. Oxygen di-negative O(-2) is pretty common but for other elements it is less common. Oxgen shows oxidation state of only –2 ( except OF2 and peroxides ). It does not show +4 and +6 oxidation states as shown by others. Most abandunt element on Earth’s crust with Si. Metals like Cu, Ag, Hg show lesser affinity with O2 to form oxide than with Sulphur to form Sulphide. ( This isdue to polarizing power of Cu(+2), Ag(+1), Hg(+2) ion. This gives strong bonds with S(-2).
!
Elements known since Antiquity -
C – Carbon – ( p – 6, M – 12 ) 17 th most abundant element. Catenation is the tendency to form long open or closed chains by the combination of same atoms. Carbon shows maximum catenation tendencies. Only Carbon can form double and triple bonds due to small size. Si, Ge, Sn and Pb are too big for the p orbitals to overlap and form bonds. Also Carbon chains can be very large while Si or Ge chains are max of 6 atoms. Graphite is prepared by Acheson process. CO2 and SiO2 are acidic while GeO2, SnO2 and PbO2 are amphoteric in nature.
!
S – Sulphur – ( p-16, M – 32 ) – Chemosynthesis is oxidation of element by bacteria. Nitrogen, Sulphur, Iron, Hydrogen and Antimony are the only elements which can undergo Chemosynthesis.
!
Au – Gold – ( p – 79, M –197 )
!
Ag – Silver – ( p – 47, M – 108 ) Argentite Ag2S Horn Silver AgCl . Siver has the higest electrical conductivity amongst all elements.
!
Cu – Copper – ( p – 29, 63.5 ) Copper Glance Cu2S Copper Pyrites CuFeS2 ( Chalcopyrites ) Malachite Cu(OH)2.CuCO3 Azurite Cu(OH)2.2CuCO3 Cuprite Cu2O
!
Fe – Iron – ( p – 26, M – 55.8 ) Ferrous Ammonium Sulphate ( Mohr’s salt ) (NH4)2SO4.FeSO4.6H2O Haematite Fe2O3 Magnetite Fe3O4 Limonite F2O3.3H2O Siderite FeCO3 . Steel rods are used in Nuclear reactors for slowing down and absorbing Neutrons. Because Iron atom is a very stable atom, and after absorbing Neutrons does not convert to Radioactive elements. Chemosynthesis is oxidation of element by bacteria. Nitrogen, Sulphur, Iron, Hydrogen and Antimony are the only elements which can undergo Chemosynthesis.
!
Pb – Lead – ( p – 82, M – 207 ) Galena PbS. Ionisation energy decreases regularly from C to Sn. Pb however shows a higher value than Sn due to poor shielding of f-orbitals. Electronegativity of Pb is higher ( or almost same ) than Sn. Pb(+2) compounds are more stable than Pb(+4) compounds. Thus Pb(+4) compounds actas oxidizing agents. Lead shows no tendency of catenation. ( The tendency to form long open or closed chains by the combination of same atoms ). Carbon shows maximum catenation tendencies. CO2 and SiO2 are acidic while GeO2, SnO2 and PbO2 are amphoteric in nature. PbCrO4 Chrome Yellow is used as Yellow Pigment. Lead Chromate PbCrO4.PbO is Red, also used as pigment. 2PbCO3.Pb(OH)2 in linseed oil is used as white paint. In electrolysis Lead cathode vanishes and gets deposited in Anode. Bismuth, Lead, Tin and Antimony settle in Anode in an eletrolysis experiment.
!
Sn – Tin – ( p – 50, M – 118.7 ) Cassiterite SnO2 . CO2 and SiO2 are acidic while GeO2, SnO2 and PbO2 are amphoteric in nature. Tin produces a peculiar sound when bent known as Tincry. Tin is not attacked by organic acids. So is used to make protection layer in cooking vessels and cooking oil containers. Solder 67% Sn 33% Pb , Bronze 75-90% Cu 25-10% Sn , Bell metal 80% Cu 20% Sn. Named from latin Stanum. Bronze swords were stronger than copper swords. Tin is used in alloy to make solders for electronic circuits / boards. In electrolysis Tin cathode vanishes and gets deposited in Anode. Bismuth, Lead, Tin and Antimony settle in Anode in an eletrolysis experiment.
!
Hg – Mercury – ( p – 80, M – 200.6 ) Alkaline solution of K2HgI4 ( Nessler’s reagent ) gives Brown precipitate with NH3 called iodide of Millon’s base ( H2NhgOHgI) Calomel Hg2Cl2 Calomel. Cinnabar HgS HgCN precipitates Pd but not other Platinum metals. Mercury boils at 357 C.
!
Elements Discovered in Middle Ages -
P – phosphorus – ( p – 15, M – 31 ) Greek Phos means Light. Phero means “ I carry “.So Phosphorus means “ I carry light “. Minerals – Phosphorite – Ca3(PO4)2 , Flourapatite – 3Ca3(PO4)2.CaF2 , Chlorapatite – 3Ca3(PO4)2.CaCl2 Nitrogen, Phosphorus, Arsenic, Antimony and Bismuth are collectively known as pnicogens. Their compounds are known as pnictides.
!
As – Arsenic – ( p – 33, M – 75 ) Nitrogen, Phosphorus, Arsenic, Antimony and Bismuth are collectively known as pnicogens. Their compounds are known as pnictides.
!
Sb – Antimony – ( p – 51, M – 122 ) Indium and Antimony together as an alloy is used for Electronics, because it is a semiconductor. Antimony is used as a catalyst to make Chloro Fluoro carbons. Nitrogen, Phosphorus, Arsenic, Antimony and Bismuth are collectively known as pnicogens. Their compounds are known as pnictides. Antimony is very toxic. Similar to Gallium and Bismuth, Antimony increases in volume when solidifies from liquid. Chemosynthesis is oxidation of element by bacteria. Nitrogen, Sulphur, Iron, Hydrogen and Antimony are the only elements which can undergo Chemosynthesis. In electrolysis Antimony cathode vanishes and gets deposited in Anode. Bismuth, Lead, Tin and Antimony settle in Anode in an eletrolysis experiment.
!
Bi – Bismuth – ( p – 83, M – 209 ) Nitrogen, Phosphorus, Arsenic, Antimony and Bismuth are collectively known as pnicogens. Their compounds are known as pnictides. Similar to Antimony and Gallium, Bismuth increases in volume when solidifies from liquid. In electrolysis Bismuth cathode vanishes and gets deposited in Anode. Bismuth, Lead, Tin and Antimony settle in Anode in an eletrolysis experiment.
!
Zn – Zinc – ( p – 30, M – 65 ) Zinc Sulphate ( White Vitriol ) ZnSO4.7H2O Zinc Blende ZnS Calamine ZnCO3 Zincite ZnO . Many enzymes in body have Zinc and is vital for the reactions to happen. Zinc helps in absobing chemicals in the body. So many medicines come along with Zinc to increase absorbtion. Zinc coated steel sheets are known as galvanised sheets, used for roofs. Zinc washers are used in underwater equipments or in boats to localize corrosion.
!
Elements Discovered by Chemical Analysis -
Co – Cobalt – ( p – 27, M – 59 ) Used to make very small powerful magnets, which are particularly used in Colour Televisions speakers, as big magnet would affect the electrons from the guns and distort the colours. Powdered Cobalt is used for catalytic reactions.
!
Ni – Nickel – ( p- 28, M – 58.7 ) Found in Canada. It is pretty toxic and has melting point within room temperature. Some people can be allergic to Nickel. Is used for plating Tin containers, steel plates etc.
!
Mn – Manganese – ( p – 25, M – 55 )
!
Ba – barium – ( p – 56, M – 137 ) Barytes BaSO4. BaO is an excellent drying agent especially for Organic bases such as Pyridine. BaO2 is used for preparation of H2O2. Ba(OH)2 is used for preparation of standard alkali solution for titrating acids. BaCl2 is used as a laboratory reagent for test of sulphate radical. Ba(ClO3)2 is used for producing Green fire and preparation of Chloric acid. Barium absorbs X-rays very well, so used for observing certain parts of body by giving the patient a Barium milk or Barium Anema ( as the case may be ). First named as Barot, then Barite from Greek Barus – Heavy. As it is a heavy Earth metal so Barium. Many types of spark plugs were made with Barium Nickel alloy.
!
Mo – Molybdenum – ( p – 42, M – 96 ) Mo is essential for life to exist as it is part of an enzyme called nitrogenase, which is responsible to absorb Nitrogen from atmosphere and convert it to proteins. A type of Bacteria that exists in the roots of beans, have Nitrogenase, convert the atmospheric Nitrogen to compounds which become part of the beans, and thus as food, become part of our body as Proteins.
!
W – Tungsten – ( p – 74, M – 184 ) – Tungsten has a boiling point close to 6000 C similar to Rhenium.
!
Te – Tellurium – ( p – 52, M – 177.6 )
!
Sr – Strontium – ( p – 38, M – 87.6 ) Mineral Celestine SrSO4, Strontianite SrCO3. Strontium 90 is radioactive which is produced in Nuclear Reaction experiments. In 1950s and 60s Strontium 90 got dispersed in the atmosphere quite widely to be ultimately seen in bones and teeth of people, resulting in lot of fear and protests. Named after a small village Strontian, in Scotland, where Strontianite mineral was found. Used in Asia for Pyroworks, and later for signaling ships. Was used to extract Sugar from molasses before Calcium started being used for the same. Radioactive Strontium-90 cells of the size of a matchbox can emit electrons which can be converted to electricity for 20 years.
!
Zr – Zirconium – ( p – 40, M – 91 ) Used for coating / cladding of Nuclear Fuel rods, used in Reactors. Used in cutting tools and instruments as it is very hard. Oxide known as Zirconia, is use for catalysts.
!
U – Uranium – ( p – 92, M – 238 )
!
Ti – Titanium – ( p – 22, M – 48 )
!
Cr – Chrimium – ( p – 24, M – 52 )
!
Be – Beryllium – ( p – 4, M – 9 ) Be is more chemically similar to Al being amphoteric than Mg – Magnesium of Group 2A. Berylium was hiding behind Alumina and was very difficult to be identified. The standard oxidation potential values of Be and Al are very close to each other. Electronegativity values are same. Mineral Beryl 3BeO.Al2O3.6SiO2 Be does not decompose in water due to low value of oxidation potential. Has tendency to form covalent compounds. Does not react directly with Hydrogen. (BeH2)n is a polymeric solid. BeCO3 is not stable towards heat. Sulphate is soluble in water due to high degree of solvation of small Be(2+) ions. Its compounds do not impart any colour in the flame, due to very high energy needed to excite the electron to higher states. It forms many chelate complexes.
!
Ni – Niobium – ( p – 28, M – 58.7 )
!
Ta – Tantalum – ( p – 73, M – 181 )
!
Elements Discovered by Electrochemical Methods –
!
Na – Sodium – ( p – 11, M – 23 ) 7 th most abundant. All Alkali elements are Silvery white Solid. Most abundant Alkali metal. Chile Saltpeter NaNO3, Glauber’s Salt Na2SO4.10H2O , Trona Na2CO3.2NaHCO3.3H2O , Natron Na2CO3.H2O , Tincal ( Borax ) Na2B4O7.10H2O , Cryolite Na3AlF6 , Soda Felspar NaAlSi3O8 Na gives Golden Yellow colour in flame test. Yellow colour of Na2O2 is probably due to presence of small amount of superoxides.
!
K – Potassium – ( p – 19, M – 39 ) 8 th most abundant. Carnallite KCl.MgCl2.6H2O , Kainite KCl.MgSO4.MgCl2.3H2O , Indian Saltpeter KNO3 , Feldspar K2O.Al2O3.6SiO2 , Polyhalite K2SO4.MgSO4.CaSO4.6H2O , Sylvine kCl All Alkali metals are paramagnetic. While their salts are diamagnetic. K gives pale violet colour in flame test.
!
Mg – Magnesium – ( p – 12, M – 24 ) Minerals – Magnesite MgCO3, Dolomite MgCO3.CaCO3, Carnallite KCl.MgCl2.6H2O, Epsomite MgSO4.7H2O, Asbestos CaMg3(SiO3)4 Kieserite MgSO4.H2O. Mg is an essential constituent of Chlorophyll.
!
Ca – Calcium – ( p – 20, M – 40 ) Gypsum CaSO4.2H2O, Fluorspar CaF2, Limestone CaCO3
Elements Predicted from Periodic Table –
Ga – Gallium – ( p – 31, M – 69.7 ) – Mendelev predicted this as Eka-Aluminium. Gallium was the first element discovered amongst the Mendelev’s more than a dozen predictions in around 1870. Named after Gallia the Latin of France. Has a low melting point 29.8 C, close to body temperature; so in some hot countries it is a liquid. This low melting point of Gallium and certain Alloys of it help making Fire alarms, so that these melt easiliy, and complete an electrical circuit to raise the alarm. The mineral Hallite in a mines of South-East Africa contains upto 37% Gallium. Coal Ash of some places are very rich in Gallium ( a few grams per tons of Ash ). Similar to Sodium it can be cut by knife. Similar to Antimony and Bismuth, Gallium grows in volume when solidifies from liquid. Gallium thermometers can be used for temperatures over 1000 C and over. ( A level Mercury thermometers cannot even approach as Mercury boils at 357 C ).
!
Sc – Scandium – ( p – 21, M – 45 ) – Mendelev had predicted Scandium as Eka-Boron from his periodic table. It is a shiny light metal. Its ability to store Hydrogen for car fuels is being explored.
!
Ge – Germanium – ( p – 32, M – 72.6 ) Named after Germany. CO2 and SiO2 are acidic while GeO2, SnO2 and PbO2 are amphoteric in nature. Ge is not attacked by dilute HCl. But when heated in HCl gas Germanium Chloroform GeHCl3 is formed. Can be used for polished mirrors.
!
The 6 Platinum metals –
!
Ru – Ruthenium – ( p – 44, M – 101 ) Named after Ruthenia ( old named of Russia ). As first element discovered by a Russian Scientist.
!
Rh – Rhodium – ( p – 45, M – 103 ) Named due to Red colour of 1st Salt produced. Greek Rodon means Rose. ( Are all Roses Red ? ). Rh is least abundant amongst Pt metals. Very widely used in Catalysts. Is very costly. Industrially Methanol ( CH3OH ) and Carbon Monoxide ( CO ) is joined togather to synthesize Ethanoic acid ( C2H5OH ) using Rhodium Catalysts.
!
Pd – Palladium – ( p – 46, M – 106.42 ) HgCN precipitates Pd but not other Platinum metals. Palladium is most abundant amongst these 6 metals but was discovered the last. Palladium was named in honour of Pallas Athena ( Greek Goddess of Wisdom, War, Civilisation ).
!
Os – Osmium – ( p – 76, M – 192 ) From Greek Osme, meaning smell. Osmium oxide is volatile and has peculiar smell.
!
Ir – Iridium – ( p – 77, M – 192 ) Got its name due to variety of colour of its salts. Greek Iris means Rainbow. The Iridium catalysts are more effective than Rhodium catalysts. So chemical engineers get more yield of compounds in the same plant using Iridium catalysts.
!
Pt – Platinum – ( p – 78, M – 195 ) The 2nd most abundant element amongst the 6 elements.
!
Elements named after Asteroids / Gods –
Pd – Palladium – ( p – 46, M – 106 ) HgCN precipitates Pd but not other Platinum metals. Palladium is most abundant amongst 6 Platinum metals but was discovered the last. Palladium was named in honour of Pallas Athena ( Greek Goddess of Wisdom, War, Civilisation ).
!
Ce – Cerium – ( p – 58, M – 140 ) Named after a “ recently “ discovered asteroid Ceres ( 1801 )
!
Elements named after colour –
Cr – Chromium – ( p – 24, M – 52 ) Greek Chroma means colour. Almost all compounds of Cr are of some colour. Siberia is the birthplace of Cr. One of the quite abundant element in the Earth’s crust. ( 0.02% of Earth’s crust by mass ). Crocoite is PbCrO4
!
Cl – Chlorine – ( p – 17, M – 35.5 ) Named after Greek Chloros meaning Yellow-Green. [ The word Halogen in Greek means Salt Producing i.e. Salt + Produce ]
!
I – Iodine – ( p – 53, M – 127 ) From Greek Iodes meaning Violet Colour.
!
Rh – Rhodium – ( p – 45, M – 103 ) Named due to Red colour of 1st Salt produced. Greek Rodon means Rose. ( Are all Roses Red ? ). Rh is least abundant amongst Pt metals.
!
Cs – Cesium – ( p – 55, M – 133 ) From Latin Caesius. Blueness of the upper part of the firmament( Celestial Sphere – The apparent surface of the imaginary sphere on which celestial bodies appear to be projected). Cs had Blue vapor. Also Blue spectral lines. All Alkali elements are Silvery white Solid. In flame test gives sky Blue colour. Melting point 28.5 C. Cesium has the best photo-electric effect, so very widely used for Photocells.
!
Rb – Rubidium – ( p – 37, M – 85.5 ) 2nd element discovered by spectroscopy. Latin Rubidius means deep red colour. Rb had red spectral lines. All Alkali elements are Silvery white Solid. In flame test gives Reddish Violet colour. Rubidium also gives out electrons very easily, but not as good as Cesium.
!
Tl – Thallium – ( p – 81, M – 204 ) 3rd element discovered by spectroscopy. Greek Thallos ( new Green Branch ) – Tl had Green spectral lines. Thallium salts are very poisonous. Thallium salts of Bromine and Iodine as a combination ( Red in coulur ) is used for making windows used in infrared experiements.
!
In – Indium – ( p – 49, M – 115 ) Extremely bright Blue spectral lines. Named after Indigo meaning Bright Blue.
!
Ir – Iridium – ( p – 77, M – 192 ) Got its name due to variety of colour of its salts. Greek Iris means Rainbow.
!
Sc – Scandium and Y – Yttrium are considered as part of “ Rare Earth elements “ along with Lanthanides. Rare Earth elements are almost 1/3 rd of the total elements, but took 113 years to discover all mainly due to rare availability and very similar chemical behavior. Y – Yttrium was the first to be discovered. Lu – Lutetium being the last. Pm – Promethium – was prepared artificially. La – Lanthanum in Greek means to lie hidden. Solid Solutions of Thorium Di-oxide and Yttrium oxide is transparent like glass for visible region. With a melting point around 2200 C this is used for peep windows for high temperature furnaces.
!
La – Lanthanum – ( p – 57, M – 139 ) Is the biggest by size amongst all Lanthanides, so most reactive. Is very soft and can be cut by a knife. La with Cu as a compound ( Lanthanum-Cuprate ) is used for catalysts. Lathanum has the property to absorb Hydrogen like a sponge and thus has application in Hydrogen fueled cars. Lanthanum has also been used for special lighting in Hollywood studios.
!
Elements named after smell -
Br – Bromine – ( p – 35, M – 80 ) Named after Greek Bromos which means stinking.
!
Os – Osmium – ( p – 76, M – 190 ) From Greek Osme, meaning smell. Osmium oxide is volatile and has peculiar smell.
!
Elements discovered by Spectroscopy –
!
Cs – Cesium – ( p – 55, M – 133 ) From Latin Caesius. Blueness of the upper part of the firmament( Celestial Sphere – The apparent surface of the imaginary sphere on which celestial bodies appear to be projected). Cs had Blue vapor. Also Blue spectral lines. 1st element established by spectroscopy. Cs is the strongest reducing agent amongst all the Alkali metals because ionization energy decreses down the group.
!
Rb – Rubidium – ( p – 37, M – 85 ) 2nd element discovered by spectroscopy. Latin Rubidius means deep red colour. Rb had red spectral lines. All the Alkali metals are Silvery white elements. Can be cut by knife except Li. Softness increases down the group due to longer bond length. In flame test the salts of Rb gives violet colour. Reducing property increases down the group, because ionization energy decreases down the group.
!
Tl – Thallium – ( p – 81, M – 204 ) 3rd element discovered by spectroscopy. Greek Thallos ( new Green Branch ) – Tl had Green spectral lines.
!
In – Indium – ( p – 49, M – 115 ) Extremely bright Blue spectral lines. Named after Indigo meaning Bright Blue.
!
He – Helium – ( p – 2, M – 4 ) New spectral lines were first observed in Sun rays ( So discovered outside Earth ). Later discovered in Hot springs and Alpha-particles trapped inside rocks as He, produced due to Radio activity.
!
Elements named after Places –
!
Y – Ytterbium – ( p – 39, M – 89 ) Ytterby is Swedish village where the mineral Ytterbite is available. Ytterbium was discovered from Ytterbite so the name !
!
Lu – Lutetium – ( p – 71, M – 175 ) Named after old name of Paris – Lutetia. Is used to date the meteorites. Used for treatment of cancer.
!
Ho – Holmium – ( p – 67, M – 165 ) Named after old name of Stockholm. It has the Highest Magnetic Moment amongst all the elements. ( Due to a large number of unpaired electrons )
!
Tm – Thulium – ( p – 69, M – 169 ) Named after a legendary country tale Thule the world’s end. Tm was so difficult to purify and discover. In beginning of 1900s Charles James, purified Thulium Chloride, by a record of 15,000 recrystalisations. So it is very expensive.
!
Se – Selenium – ( p – 34, M – 79 ) Se was discovered late because it was hiding behind S ( Sulphur ) and Te ( Telurium ). Selenium is named after Greek Selenus meaning moon.
!
Radioactive Elements –
!
Po – Polonium – ( p – 84, M – 209 ) ( Radioactive )
!
Ra – Radium – ( p – 88, M – 226 ) ( Radioactive )
!
Ac – Actinium – ( p – 89, M – 227 ) ( Radioactive ) The actinides have been named after Actinium. Actinium glows with a light bluish tinge in dark. It is more lethal and toxic than Polonium which was considered to be most lethal once upon a time.
!
Rn – Radon – ( p – 86 , M – 222 ) ( Radioactive ) Radon is produced by Radioactive decomposition of other elements. So houses made above Granite deposites often contain more than average level of Radon. This Radioactive gas can cause lung cancer.
!
Pa – Protactinium – ( p – 91, M – 231 ) ( Radioactive ) Only 125 kg was purified from 60 tons of nuclear waste after 12 costly steps. This being radioactive, has not much of use.
!
Fr – Francium – ( p – 87, M – 223 ) ( Radioactive )
!
Rf – Rutherfordium – ( p – 104, M – 261 ) – ( Radioactive ) Rutherford in 1919 bombarded Nitrogen with Alpha particles to produce some Oxygen atoms. This was amongst the first examples of transformation of elements.
!
Synthesized Elements –
!
Tc – Technetium – ( p – 43, M – 98 ) ( Radioactive ) The first element in the periodic table to be radioactive.
!
Pm – Promethium – ( p – 61, M – 145 ) ( Radioactive ) The only Lanthanide which is radioactive. So its chemistry is hardly studied.
!
At – Astatine – ( p – 85, M – 210 ) ( Radioactive ) No particular use, as it is radioactive. The atoms and the compounds decay very fast, so chemists could not study Astatine much.
!
Fr – Francium – ( p – 87, M – 223 ) ( Radioactive )
!
Transuranium Elements –
Np – Neptunium – ( p – 93, M – 237 ) ( Radioacive ) Was the first synthetic element. Also found in traces in Nuclear Bomb experiement sites.
!
Pu – Plutonium – ( p – 94, M – 244 ) ( Radioactive )
!
Am – Americium – ( p – 95, M – 243 ) ( Radioactive ) The only synthetic elements used very widely in households. This is part of the “ smoke Detectors “, due to its ability to detect various ions in air, as it happens in case of smoke.
!
Cm – Curium – ( p – 96, M – 247 ) ( Radioactive ) Named after Marie Curie and Pierre Curie. Can be used for thermoelectric generators, but practical / commercial use is shunned because highly radioactive and has be produced artificially.
!
Bk – Berkelium – ( p – 97, M – 247 ) A 3 billionth of a gram was made in Berkeley, and was just enough to observe some spectroscopical data.
!
Cf – Californium – ( p – 98, M – 251 ) ( Radioactive ) It is used to detect metal fatigue and stress, as the instruments for observing these have Californium. Also used in metal detectors. Similar to Americium it is commonly used in household appliances.
!
Es – Einsteinium – ( p – 99, M – 252 ) ( Radioactive ) First found in the debri site of an Nuclear Bomb experiment.
!
Fm – Fermium – ( p – 100, M – 257 ) ( Radioactive )
!
Md – Mendelevium – ( p – 101, M – 258 ) ( Radioactive )
!
No –Nobelium – ( p – 102, M – 259 ) ( Radioactive )
!
Bh – Bohrium – ( p – 107, M – 262 ) ( Radioactive )
!
Db – Dubnium – ( p – 105, M – 262 ) ( Radioactive ) Named after Russian atomic research centre at Dubna.
!
Uuu – Unununium – Rg – Roentgenium – ( p – 111, M – 272 ) Artificially made. Named after Roentgen the 1st Nobel Laureate in Chemistry.
!
Uub – Ununbium – ( p – 112, M – ) Copernicium was synthesized in 1996 at GSI
!
Uut – Ununtrium – ( p – 113, M – ) Sythesized in accelerator facility at RIKEN, Japan.
!
Uup – Ununpentium – ( p – 115 , M – ) Made artificially. Belongs to VA group.
!
Uus – Ununseptium – ( p – 117, M – ? ) Very difficult to synthesize as 117 proton number seems to be highly unstable.
!
B – Boron – ( p – 5, M – 11 ) Boron has maximum covalency of 4 due to non-availability of d-electrons. While all other elements of Group 3A have max covalency as 6.
Boron alone exhibits allotropy in the group. Boron shows +3 oxidation state while others in 3A shows +1 and +3. Boron does not form cations in aqueous solutions as the hydration energy is less than sum of the 3 successive ionization energies. Boron halides are monomeric while other halides of 3A are dimeric. Boron does not show iner pair effect. Boron forms a number of volatile hydrides which are electron deficient compounds. Others form only one polymeric hydride. Thallium does not form hydride. Boron does not decompose water or steam, while other elements of 3A decompose water or steam. B2O3 is acidic in nature. Other oxides of 3A are either amphoteric or basic. B(OH)3 is a weak acid, Al(OH)3 is amphoteric in nature. Boron is not attacked by non-oxidising acids, while other elements are attacked. Boron dissolves in conc. HNO3 forming H3BO3. The other elements become passive esp. Al, Ga. Boron combines with metals and form borides while other elements do not combine. They can form alloys. BCl3 is fuming liquid, while AlCl3 is a solid.
Minerals – Borax (Tincal ) Na2B4O7.10H2O Colemanite Ca2B6O11 5H2O Panderinite Ca2B6O11.3H2O Boracite 2Mg3B8O15.MgCl2 Boronatro Calcite CaB4O7.NaBO2.8H2O Kernite ( Rasorite ) Na2B4O7.4H2O Boric Acid H3BO3
Boron Banana Bond
B2O3 – Boron Trioxide is also called Sesquioxide. Borax – Sodium Tetraborate Na2B4O7.10H2O
!
N – Nitrogen – ( p – 7, M – 14 ) Nitrogen is a gas while other members of Group 5A are solids. Catenation property of N is known. Chains upto 8 Nitrogen atoms are known. Chile Saltpeter NaNO3 Indian saltpeter KNO3 . Chemosynthesis is oxidation of element by bacteria. Nitrogen, Sulphur, Iron, Hydrogen and Antimony are the only elements which can undergo Chemosynthesis.
!
Pr – Praseodymium – ( p – 59, M – 141 ) Praseodymium has lots of uses. Added in some types of Silicates, produces a material with very high refractive index that can reduce the speed of light to mere 300 m/sec . Also used in the coils, that were used to produce very low temperature close to 0 Kelvin. ( 1 billionth of a degree away from Zero kelvin ). Is used in welder’s goggles to filter out harmful UV lights.
!
Kr – Krypton – ( p – 36, M – 83 ) Can make compounds with Fluorine. A very high energy UV light is needed to break the F-F bond and make Kr Fluoride. Kr Fluoride is used to make very high energy UV pulse LASERS.
!
Eu – Europium – ( p – 63, M – 152 ) This Lanthanide is not radioactive and its salts are used for getting various colours in the Picture / cathode ray tube when bombarded by electron guns. Europium salts were particularly used for getting Red and Blue colour in the beginning of colour television sets.
!
Tb – Terbium – ( p – 65, M – 159 ) Similar to Europium the salts of Terbium is used to give Green and Yellow colours in the Picture / cathode ray tube when bombarded by electron guns. Used as a marker in Biochemistry experiments due to its fluorescence nature. In certain magnetic materials, which contain Terbium the size changes with direction of Magnetic field, giving rise to many technical applications.
!
Sm – Samarium – ( p – 62, M – 150 ) Samarium is known to change its oxidation states very easily and thus has quite a few uses. It is a good Neutron absorber so has uses in Nuclear power plants for slowing down and controlling the Nuclear reactions. Samarium is used to date the old samples of rocks such as moon rocks.
!
Ar – Argon – ( p – 18, M – 40 ) Nearly 1% of Earth’s Atmosphere is Argon ( 0.93% ). It is widely used for packaging pure elements and compounds to avoid oxidation by atmospheric Oxygen. Purged and blown around liquid metals, such as Aluminium or Iron to stop action of Oxygen, Nitrogen etc. Neon lights made of Argon gives nice Blue colour.
!
Dy – Dysprosium – ( p – 66, M – 162.5 ) This lanthanide is very good in absorbing Neutrons. So used as coatings of Nuclear rods and control the Nuclear reactions. When mixed with Sulphur and Cadmium can produce Infrared when irradiated with EM waves. This Infrared is used to study structure of molecules by studying the absorption spectra.
!
Cd – Cadmium – ( p – 48, M – 112 ) Used in batteries. As a metal it is toxic but various compounds of Cadmium are used for colouring, such as Plastic pipes. Cadmium Sulfide has Orange colour and commonly used as a dye. Cadmium was named after Cadmea in Greek which meant Zinc Ore. An alloy of 99% Cd and 1% Ni is used in bearings as antifriction.
Xanthate
Xanthate usually refers to a salt with the formula ROCS−2M+ (R = alkyl; M+ = Na+, K+). The name xanthates is derived from Greek ξανθός xanthos, meaning “yellowish, golden”, and indeed most xanthate salts are yellow. They were discovered and named in 1823 by the Danish chemist William Christopher Zeise. These organosulfur compounds are important in two areas, the production of cellophane and related polymers from cellulose and secondly in mining for the extraction of certain ores. They are also versatile intermediates in organic synthesis. Xanthates also refer to esters of xanthic acid. These esters have the structure ROC(=S)SR′.
Certain xanthate salts and bisxanthates (e.g. Dixanthogen) are used as flotation agents in mineral processing. They are intermediates in the Chugaev elimination process and are used to control radical polymerisation under the RAFT process, also termed MADIX (macromolecular design via interchange of xanthates).
Xanthate salts are produced by the reaction of an alcohol with sodium or potassium hydroxide and carbon disulfide:
ROH + CS2 + KOH → ROCS2K + H2O
The reaction involves the attack of the alkoxide salt. For example, sodium ethoxide gives sodium ethyl xanthate. Virtually any alcohol can be used in this reaction. Technical grade xanthate salts are usually of 90–95% purity. Impurities include alkali-metal sulfides, sulfates, trithiocarbonates, thiosulfates, sulfites, or carbonates as well as residual raw material such as alcohol and alkali hydroxide. These salts are available commercially as powder, granules, flakes, sticks, and solutions are available. China is a major exporter of granules.
Some commercially important xanthate salts include:
sodium ethyl xanthate CH3CH2OCS2Na
potassium ethyl xanthate, CH3CH2OCS2K
sodium isopropyl xanthate, (CH3)2CHOCS2Na
sodium isobutyl xanthate, (CH3)2CHCH2OCS2Na
potassium amyl xanthate, CH3(CH2)4OCS2K
The OCS2 core of xanthate salts and esters is characteristically planar. The central carbon is sp2-hybridized.
Xanthate salts characteristically decompose in acid:
ROCS2K + HCl → ROH + CS2 + KCl
This reaction is the reverse of the method for the preparation of the xanthate salts. The intermediate in the decomposition is the xanthic acid, ROC(S)SH, which can be isolated in certain cases.
Xanthate anions also undergo alkylation to give xanthate esters, which are generally stable:
ROCS2K + R′X → ROC(S)SR′ + KX
The C-O bond in these compounds are susceptible to cleavage by the Barton–McCombie deoxygenation, which provides a means for deoxygenation of alcohols.
They can be oxidized to the so-called dixanthogens:
2 ROCS2Na + Cl2 → ROC(S)S2C(S)OR + 2 NaCl
Xanthates bind to transition metal cations as bidentate ligands. The charge-neutral complexes are soluble in organic solvents.
Industrial applications
Formation of sodium cellulose xanthate starting from cellulose
Cellulose reacts with carbon disulfide (CS2) in presence of sodium hydroxide (NaOH) to produces sodium cellulose xanthate, which upon neutralization with sulfuric acid (H2SO4) gives viscose rayon or cellophane paper (Sellotape or Scotch Tape).
Certain xanthate salts and bisxanthates (e.g. Dixanthogen) are used as flotation agents in mineral processing. They are intermediates in the Chugaev elimination process and are used to control radical polymerisation under the RAFT process, also termed MADIX (macromolecular design via interchange of xanthates).
Clathrate compound
A clathrate is a chemical substance consisting of a lattice that traps or contains molecules. The word clathrate is derived from the Latin clatratus meaning with bars or a lattice. Traditionally, clathrate compounds are polymeric and completely envelop the guest molecule, but in modern usage clathrates also include host-guest complexes and inclusion compounds. According to IUPAC, clathrates are "Inclusion compounds in which the guest molecule is in a cage formed by the host molecule or by a lattice of host molecules."
Traditionally clathrate compounds refer to polymeric hosts containing molecular guests. More recently, the term refers to many molecular hosts, including calixarenes and cyclodextrins and even some inorganic polymers such as zeolites. The natural silica clathrate mineral, chibaite was recently described from Japan.
Many clathrates are derived from organic hydrogen-bonded frameworks. These frameworks are prepared from molecules that "self-associate" by multiple hydrogen-bonding interactions. The most famous clathrates are methane clathrates where the hydrogen-bonded framework is contributed by water and the guest molecules are methane. Large amounts of methane naturally frozen in this form exist both in permafrost formations and under the ocean sea-bed. Other hydrogen-bonded networks are derived from hydroquinone, urea, and thiourea. A much studied host molecule is Dianin's compound.
Structure of the clathrate consisting of cadmium cyanide host and carbon tetrachloride guest,Cd(CN)2.CCl4. Blue = Cd(CN)2 framework, gray = C, green = disordered Cl.
Hofmann compounds are coordination polymers with the formula Ni(CN)4Ni(NH3)2. These materials crystallize with small aromatic guests (benzene, certain xylenes), and this selectivity has been exploited commercially for the separation of these hydrocarbons. Metal organic frameworks (MOFs) form clathrates.
Example of Metal organic framework, MOF-5, the cavity for the guests is indicated by the yellow sphere.
Photolytically-sensitive caged compounds have been examined as containers for releasing a drug or reagent.
Clathrate hydrates were discovered in 1810 by Humphry Davy. Clathrates were studied by P. Pfeiffer in 1927 and in 1930, E. Hertel defined "molecular compounds" as substances decomposed into individual components following the mass action law in solution or gas state. In 1945, H. M. Powell analyzed the crystal structure of these compounds and named them clathrates.
Chalcogen
The chalcogens (/ˈkælkədʒᵻnz/) are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. It consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive element polonium (Po). The chemically uncharacterized synthetic element livermorium (Lv) is predicted to be a chalcogen as well. Often, oxygen is treated separately from the other chalcogens, sometimes even excluded from the scope of the term "chalcogen" altogether, due to its very different chemical behavior from sulfur, selenium, tellurium, and polonium. The word "chalcogen" is derived from a combination of the Greek word khalkόs (χαλκός) principally meaning copper (the term was also used for bronze/brass, any metal in the poetic sense, ore or coin), and the Latinised Greek word genēs, meaning born or produced.
Sulfur has been known since antiquity, and oxygen was recognized as an element in the 18th century. Selenium, tellurium and polonium were discovered in the 19th century, and livermorium in 2000. All of the chalcogens have six valence electrons, leaving them two electrons short of a full outer shell. Their most common oxidation states are −2, +2, +4, and +6. They have relatively low atomic radii, especially the lighter ones.
Lighter chalcogens are typically nontoxic in their elemental form, and are often critical to life, while the heavier chalcogens are typically toxic. All of the chalcogens have some role in biological functions, either as a nutrient or a toxin. The lighter chalcogens, such as oxygen and sulfur, are rarely toxic and usually helpful in their pure form. Selenium is an important nutrient but is also commonly toxic. Tellurium often has unpleasant effects (although some organisms can use it), and polonium is always extremely harmful, both in its chemical toxicity and its radioactivity.
Sulfur has more than 20 allotropes, oxygen has nine, selenium has at least five, polonium has two, and only one crystal structure of tellurium has so far been discovered. There are numerous organic chalcogen compounds. Not counting oxygen, organic sulfur compounds are generally the most common, followed by organic selenium compounds and organic tellurium compounds. This trend also occurs with chalcogen pnictides and compounds containing chalcogens and carbon group elements.
Oxygen is generally extracted from air and sulfur is extracted from oil and natural gas. Selenium and tellurium are produced as byproducts of copper refining. Polonium and livermorium are most available in particle accelerators. The primary use of elemental oxygen is in steelmaking. Sulfur is mostly converted into sulfuric acid, which is heavily used in the chemical industry. Selenium's most common application is glassmaking. Tellurium compounds are mostly used in optical disks, electronic devices, and solar cells. Some of polonium's applications are due to its radioactivity.
Pnictogen
A pnictogen is a member of the nitrogen group of elements, Group 15 of the periodic table (formerly numbered as Group V or Group VA). This group consists of nitrogen, phosphorus, arsenic, antimony, bismuth, and ununpentium. The pnictogens are noted for their ability to form stable compounds, thanks to their tendency to form double and triple covalent bonds. The pnictogens are solids at room temperature, except for nitrogen, which is a gas. This group is also known as the nitrogen family. It consists of the elements nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), and perhaps also the chemically uncharacterized synthetic element moscovium (Mc).
The name pentels (from Greek πέντε, pénte, five) was also used for this group at one time.
The defining characteristic of pnictogens is that atoms of these elements have 5 electrons in their outer electron shell. There are 2 paired electrons in the s subshell and 3 unpaired electrons in the p subshell, putting these elements 3 electrons shy of filling the outermost shell.
Binary compounds from this group are termed pnictides.
Pnictide compounds tend to be exotic. Various properties that some pnictides have include being diamagnetic and paramagnetic at room temperature, being transparent, and generating electricity when heated. Other pnictides include the ternary rare-earth main-group variety of pnictides. These are in the form of REaMbPnc, where M is a carbon group or boron group element and Pn is any pnictogen except nitrogen. These compounds are between ionic and covalent compounds and thus have unusual bonding properties.
These elements are also noted for their stability in compounds due to their tendency for forming double and triple covalent bonds. This is the property of these elements which leads to their potential toxicity, most evident in phosphorus, arsenic and antimony. When these substances react with various chemicals of the body, they create strong free radicals not easily processed by the liver, where they accumulate. Paradoxically it is this strong bonding which causes nitrogen and bismuth's reduced toxicity (when in molecules), as these form strong bonds with other atoms which are difficult to split, creating very unreactive molecules. For example, N2, the diatomic form of nitrogen, is used as an inert gas in situations where using argon or another noble gas would be too expensive.
The upper pnictogens, that is, nitrogen, phosphorus, and arsenic tend to form −3 charges. Antimony and bismuth can either take on a +3 or +5, by losing its p-shell electrons or losing its p-shell and s-shell electrons, respectively.
Pnictogens can react with hydrogen to form pnictogen hydrides such as ammonia.
A pnictogen is a member of the nitrogen group of elements, Group 15 of the periodic table (formerly numbered as Group V or Group VA). This group consists of nitrogen, phosphorus, arsenic, antimony, bismuth, and ununpentium. The pnictogens are noted for their ability to form stable compounds, thanks to their tendency to form double and triple covalent bonds. The pnictogens are solids at room temperature, except for nitrogen, which is a gas. This group is also known as the nitrogen family. It consists of the elements nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), and perhaps also the chemically uncharacterized synthetic element moscovium (Mc).
The name pentels (from Greek πέντε, pénte, five) was also used for this group at one time.
The defining characteristic of pnictogens is that atoms of these elements have 5 electrons in their outer electron shell. There are 2 paired electrons in the s subshell and 3 unpaired electrons in the p subshell, putting these elements 3 electrons shy of filling the outermost shell.
Binary compounds from this group are termed pnictides.
Pnictide compounds tend to be exotic. Various properties that some pnictides have include being diamagnetic and paramagnetic at room temperature, being transparent, and generating electricity when heated. Other pnictides include the ternary rare-earth main-group variety of pnictides. These are in the form of REaMbPnc, where M is a carbon group or boron group element and Pn is any pnictogen except nitrogen. These compounds are between ionic and covalent compounds and thus have unusual bonding properties.
These elements are also noted for their stability in compounds due to their tendency for forming double and triple covalent bonds. This is the property of these elements which leads to their potential toxicity, most evident in phosphorus, arsenic and antimony. When these substances react with various chemicals of the body, they create strong free radicals not easily processed by the liver, where they accumulate. Paradoxically it is this strong bonding which causes nitrogen and bismuth's reduced toxicity (when in molecules), as these form strong bonds with other atoms which are difficult to split, creating very unreactive molecules. For example, N2, the diatomic form of nitrogen, is used as an inert gas in situations where using argon or another noble gas would be too expensive.
The upper pnictogens, that is, nitrogen, phosphorus, and arsenic tend to form −3 charges. Antimony and bismuth can either take on a +3 or +5, by losing its p-shell electrons or losing its p-shell and s-shell electrons, respectively.
Pnictogens can react with hydrogen to form pnictogen hydrides such as ammonia.
Interstitial defect
Interstitials defects are a variety of crystallographic defects where atoms assume a normally unoccupied site in the crystal structure. In interstitial defects two or more atoms may share one lattice site, thereby increasing its total energy. Alternatively small atoms in some crystals may occupy interstitial sites in energetically favorable configurations, such as hydrogen in palladium. Interstitials can be produced by bombarding a crystal with elementary particles having energy above the displacement threshold for that crystal, but they may also exist in small concentrations in thermodynamic equilibrium.
Impurity interstitials
Small impurity interstitial atoms are usually on true off-lattice sites between the lattice atoms. Such sites can be characterized by the symmetry of the interstitial atom position with respect to its nearest lattice atoms. For instance, an impurity atom I with 4 nearest lattice atom A neighbours (at equal distances) in an fcc lattice is in a tetrahedral symmetry position, and thus can be called a tetrahedral interstitial.
Large impurity interstitials can also be in split interstitial configurations together with a lattice atom, similar to those of the self-interstitial atom.
Interstitials defects are a variety of crystallographic defects where atoms assume a normally unoccupied site in the crystal structure. In interstitial defects two or more atoms may share one lattice site, thereby increasing its total energy. Alternatively small atoms in some crystals may occupy interstitial sites in energetically favorable configurations, such as hydrogen in palladium. Interstitials can be produced by bombarding a crystal with elementary particles having energy above the displacement threshold for that crystal, but they may also exist in small concentrations in thermodynamic equilibrium.
Impurity interstitials
Small impurity interstitial atoms are usually on true off-lattice sites between the lattice atoms. Such sites can be characterized by the symmetry of the interstitial atom position with respect to its nearest lattice atoms. For instance, an impurity atom I with 4 nearest lattice atom A neighbours (at equal distances) in an fcc lattice is in a tetrahedral symmetry position, and thus can be called a tetrahedral interstitial.
Large impurity interstitials can also be in split interstitial configurations together with a lattice atom, similar to those of the self-interstitial atom.
Pseudohalides
The pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds. Pseudohalogens occur in pseudohalogen molecules, inorganic molecules of the general forms Ps–Ps or Ps–X (where Ps is a pseudohalogen group), such as cyanogen; pseudohalide anions, such as cyanide ion; inorganic acids, such as hydrogen cyanide; as ligands in coordination complexes, such as ferricyanide; and as functional groups in organic molecules, such as the nitrile group. Well-known pseudohalogen functional groups include cyanide, cyanate, thiocyanate, and azide.
Many pseudohalogens are known by specialized common names according to where they occur in a compound. Well-known ones include
Examples of pseudohalogen molecules
Examples of symmetrical pseudohalogens (Ps–Ps) include cyanogen (CN)2, thiocyanogen (SCN)2, selenorhodane (SeCN)2, azidodithiocarbonate (N3CS2)2. Another complex symmetrical pseudohalogen is dicobalt octacarbonyl, Co2(CO)8. This substance can be considered as a dimer of the hypothetical cobalt tetracarbonyl, Co(CO)4.
Examples of non-symmetrical pseudohalogens (Ps–X), analogous to the binary interhalogen compounds, are cyanogen halides (ICN, ClCN, BrCN), and other compounds. Sometimes nitrosyl chloride NOCl also is considered as pseudohalogen.
Not all combinations are known to be stable.
-
Pseudohalides
Pseudohalides are the anions (or functional groups) of corresponding pseudohalogen groups such as cyanides, cyanates, isocyanates, rhodanides (i.e. thiocyanates and isothiocyanates), selenocyanogens, tellurorhodanides and azides.
A common complex pseudohalide is tetracarbonylcobaltate (Co(CO)4−). The acid HCo(CO)4 is in fact quite a strong acid, though its low solubility renders it not as strong as the true hydrohalic acids.
The behavior and chemical properties of the above pseudohalides are identical to that of the true halide ions. The presence of the internal double bonds or triple bonds do not appear to affect their chemical behavior. For example, they can form strong acids of the type HX (compare HCl to HCo(CO)4), and they can react with metals to form compounds like MX (compare NaCl to NaN3).
Nanoclusters of aluminium (often referred to as superatoms) are sometimes considered to be pseudohalides since they, too, behave chemically as halide ions, forming Al13I2− (analogous to I3−) and similar compounds. This is due to the effects of metallic bonding on small scales.
The N3- ( The ion N3 minus ) is known to be weak lewis base.
Why does Manganese have a low melting and boiling point ?
The melting and boiling points of transition elements increases from scandium (1530 ∘C ) to vanadium (1917 ∘C ). They increase because as we go across the period, we have more unpaired (free) electrons. Melting point of Chromium (1890 ∘C). Chromium is remarkable for its magnetic properties: it is the only elemental solid which shows antiferromagnetic ordering at room temperature (and below). Above 38 °C, it changes to paramagnetic.
The graph of melting points and boiling points is given below
In both Mn and Zn: the filled s-orbitals and ( half ) filled d-orbitals impact the bonding character of the elements. This is also apparent in the 2nd and 3rd row transition metals, though to a lesser extent due to more diffuse orbitals, greater shielding of the valence electrons and relativistic effects from heavier nuclei.
Manganese has a half-filled 3d shell ( 5 electrons in 5 degenerate orbitals ). This imparts an added stabilization to those 5 electrons since the atom as a whole has no angular momentum, because it is spherically symmetric. Similar phenomena take place for Technetium and Rhenium. Zinc, Cadmium and Mercury experience a similar but more pronounced stabilization due to a full d shell which experiences not just a stabilization; due to spherical symmetry (To put it simply) but also because all of their electrons are paired up. All of the elements just listed also experience an increase in ionization energy since going from a 5 to a 4-electron system would result in a more significant destabilization as compared to going from, say, a 3 to a 2-electron system. Even more evidence for the stabilization in the 5-electron case comes from the fact that Chromium, the element that comes before manganese has the electron configuration 4s1 3d5.
In other words, the 5 electrons of Mn are more okay with being unpaired than, say, Cr or Fe. They don't want to form the metal-metal bonds that other metals form that make them solid/liquid, as much.
In general, half-filled shells experience a stabilization. The s-orbitals are a trivial case, since we can't really measure the stabilization relative to a "quarter filled S orbital", but p-shells also experience a stabilization when you hit 3 p-electrons, and f when you hit 7.
Manganese has a half-filled 3d shell ( 5 electrons in 5 degenerate orbitals ). This imparts an added stabilization to those 5 electrons since the atom as a whole has no angular momentum, because it is spherically symmetric. Similar phenomena take place for Technetium and Rhenium. Zinc, Cadmium and Mercury experience a similar but more pronounced stabilization due to a full d shell which experiences not just a stabilization; due to spherical symmetry (To put it simply) but also because all of their electrons are paired up. All of the elements just listed also experience an increase in ionization energy since going from a 5 to a 4-electron system would result in a more significant destabilization as compared to going from, say, a 3 to a 2-electron system. Even more evidence for the stabilization in the 5-electron case comes from the fact that Chromium, the element that comes before manganese has the electron configuration 4s1 3d5.
In other words, the 5 electrons of Mn are more okay with being unpaired than, say, Cr or Fe. They don't want to form the metal-metal bonds that other metals form that make them solid/liquid, as much.
In general, half-filled shells experience a stabilization. The s-orbitals are a trivial case, since we can't really measure the stabilization relative to a "quarter filled S orbital", but p-shells also experience a stabilization when you hit 3 p-electrons, and f when you hit 7.
From the physicists' "electron sea" point of view of metal bonding, the higher the ionic charge the metal atom can support, the higher the element's melting and boiling points. This explains why group 1 metals such as sodium have quite low melting/boiling points, since the metal would be composed of electrons delocalized in a M+ lattice. Going towards group 2 and group 3 elements, one can expect to find a M2+ and M3+ lattice, and so on. However, this does not mean that that, for example, metallic Osmium is expected to be made out of Os8+ cations surrounded by a dense electron sea; as each successive "ionization" of the metal atom is performed, it becomes more unlikely that the next electron can become delocalized, since it has to fight against a higher effective nuclear charge relative to the electron before it. Therefore, we can expect some "optimal average charge" for the metal ions in the pure metal. Since melting and boiling points tend to increase towards groups 7 and 8, we can safely assume that this optimal average charge is at least greater than +3, and it is likely higher for the heavier elements since they have significantly higher melting/boiling points.
We can somehow figure out how easily electrons can be removed from their parent atoms to create the electron sea, we may have an argument to explain the trends and apparent discontinuities. First, notice that the 4th4th period metals are in general a little bit underwhelming compared their 5th5th and 6thth period counterparts when looking at the boiling points. This can be attributed to the fact that the 3d orbitals are anomalously compact relative to higher nd orbitals. This happens because all first occurrences of a given sublevel (i.e. 1s, 2p, 3d, 4f, 5g, etc.) are composed of orbitals whose wavefunctions have no radial node. Since the wavefunction's value is monotonically increasing up to its maximum value, it tapers off quickly. Thus, when a 4th period metal atom tries to delocalize its 3d electrons, it meets a slight increase in resistance due to its stronger interaction with the nucleus, meaning that the optimal average charge of the ions is slightly lower and leading to lesser interatomic interactions holding the metal together.
Now, for the sudden dips, most visible for Cr, Mn and Tc. As has been suggested, one is tempted to take into consideration the fact that they all have d5 configurations, which represent a half-filled d subshell. It can be argued that they are particularly stable due to exchange energy, though Cann suggests it is not the most adequate explanation and instead forwards the "parallel spin avoidance factor".
In half-filled subshell configurations, there is a maximum in the effective nuclear charge felt by the electrons (compared to the previous elements with no doubly-occupied orbitals) combined with relatively low interorbital repulsions due to the Pauli exclusion principle. The element that follows a half-filled subshell must now put an electron in a previously occupied orbital, creating stronger, intraorbital electron repulsion. These effects combine to suggest that removing an electron from or forcing an electron into half-filled configurations is particularly difficult ( notice that Cr doesn't follow Mn and Tc ). If this is the case, then the delocalization of the third electron ( which would create Mn3 and Tc3+ ions ) and all those beyond it would be somewhat suppressed. This would effectively reduce the optimal average charge of the ions in the metal, and decrease the strength of interactions.
Finally, Cr doesn't show the same electronic behaviour as Mn and Tc, even though it has a d5 configuration, and yet it is still anomalous. According to Greenwood and Earnshaw (Chemistry of the Elements, 1997), the low boiling point of Cr is due to a combination of increased effective nuclear charge and the anomalously small size of 3d orbitals. When going from V to Cr the increase in nuclear charge is enough to pull the already small 3d orbitals quite close to the nucleus, disfavouring electron delocalization to the extent that a sudden drop appears. Mo has the same electron configuration as Cr yet it does not show nearly as large a dip because its 4d orbitals are already significantly more diffuse, and aren't so easily suppressed from delocalizing.